Atomic weight
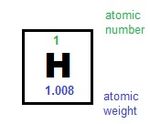
Figure 1. The atomic weight of an element is shown on the periodic table below its abbreviation.[1]
Atomic weight, or relative atomic mass, is a unitless number defined as the ratio of the average atomic mass of a given element to the atomic mass unit.[2] When multiplied with the molar mass constant, 1 g/mol, the atomic weight gives the molar mass of an element or molecule.
Please visit the page on molar mass for more information.
For Further Reading
- Atomic mass
- Molecule
- Mole
- Element
- Or explore a random page
References
- ↑ Wikimedia commons [Online], Available: http://commons.wikimedia.org/wiki/File:Atomic_number_Atomic_weight.jpg
- ↑ IUPAC, relative atomic mass (atomic weight) Ar [Online], Available: http://goldbook.iupac.org/R05258.html

