Beta decay

Beta decay is a nuclear decay process where an unstable nucleus transmutes and ejects particles to become more stable. There are two different types of beta decay - beta minus and beta plus. In both of these decays, a nucleon in the nucleus is transformed into a different type of nucleon, releasing particles in the process. Both beta minus and beta plus decay are moderately penetrating (ie the radiation can go deep inside a solid object). There's a closely related process called electron capture, where an electron is captured in the nucleus which acts just like beta plus.
Beta minus decay occurs whenever a nucleus has too many neutrons. In this type, a neutron from the nucleus is transformed into a proton and an electron, with the electron being ejected from the nucleus. To ensure the rules of particle physics hold, a tiny particle known as an anti-neutrino is also released.[2] The general equation representing beta minus decay is:
where:
- is the parent nucleus
- is the daughter nucleus
- is the released beta particle, an electron
- is the released anti-neutrino
Beta plus decay comes from a nucleus with too many protons. In this type of decay, a proton from the nucleus is transformed into a neutron and a positron (which is simply a "positive version" of the electron). To ensure rules of particle physics hold, a tiny particle known as a neutrino is also released.[2] The general equation representing beta positive decay is:
where:
- is the parent nucleus
- is the daughter nucleus
- is the released beta particle, a positron
- is the released neutrino
In both beta minus and beta plus decay it is the weak nuclear force that results in the changing of a nucleon into a different nucleon.
Safety
Beta radiation is slightly more penetrating than alpha radiation, but still not nearly as penetrating as gamma radiation. Generally speaking, because beta radiation isn't extremely penetrating it is mainly an issue when ingested. If a beta source enters the body, it causes tissue damage and can increase the risk of cancer.[3] Figure 2 shows the relative levels of penetration of a variety of different radiation types.
Exposure to beta radiation can cause a wide variety of health effects. Generally speaking, exposure to beta decay sources are chronic in nature. Chronic effects are the result of low-level exposures to beta radiation over an extended period of time, and can take anywhere between 5 and 30 years to develop.[3] The most prominent side effect of exposure is cancer. Some beta emitters are distributed throughout the body - such as carbon-14 (which occurs naturally at levels that cause no harm to the human body)- while others accumulate in specific organs. An example of this would be iodine-131, which concentrates in the thyroid gland and increases the risk of thyroid cancer.[3]
Applications and Importance
Elements that have beta decay can have useful medical applications. Radionuclide therapy (RNT) or radiotherapy is a cancer treatment that uses beta decay. In this process, lutetium-177 or yttrium-90 is attached to a molecule and ingested.[6] Once inside the body, this molecule travels to the cancer cells. The radioactive atoms then undergo a decay process, releasing beta particles and killing nearby cancer cells.
Additionally, carbon dating relies on the properties of beta decay. To determine the approximate age of artifacts, wood, and animal remains the ratio of carbon-14 to carbon-12 in an object must be determined.[6] Carbon-14 is generated from sunlight in the atmosphere from nitrogen-14, which plants breathe in with photosynthesis, and thus there is a certain amount of carbon-14 in organic remains. Plants are eaten by animals and get carbon-14 as well. When an organic body begins to decay, some of this carbon-14 becomes nitrogen-14 (through a beta decay process), and over the years the amount of carbon-14 in the sample is depleted.[6] By looking at the ratio of carbon-14 to carbon-12, the approximate age of the artifact can be determined.
To learn more about beta decay please see hyperphysics.
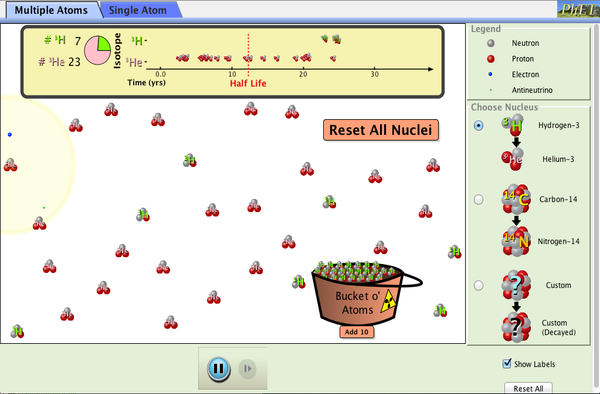
PhET
The University of Colorado has graciously allowed us to use the following PhET simulation. This simulation illustrates how radioactive nuclei decay through beta decay, and shows the half-life of these atoms.
References
- ↑ Wikimedia Commons. (July 22, 2015). Beta Minus Decay [Online]. Available: https://upload.wikimedia.org/wikipedia/commons/thumb/a/aa/Beta-minus_Decay.svg/1024px-Beta-minus_Decay.svg.png
- ↑ 2.0 2.1 Study Physics. (July 22, 2015). Beta Decay [Online]. Available: http://www.studyphysics.ca/2007/30/08_atomic/43_decay.pdf
- ↑ 3.0 3.1 3.2 US EPA. (July 22, 2015). Beta Particles [Online]. Available: http://www.epa.gov/radiation/understand/beta.html#healtheffects
- ↑ Chubu Electric Power. (May 26, 2015). Characteristics of radiation and radioactivity [Online]. Available: http://hamaoka.chuden.jp/english/radioactivity/aspect.html
- ↑ Created internally by a member of the Energy Education team.
- ↑ 6.0 6.1 6.2 ChemTeacher. (July 22, 2015). Beta Decay [Online]. Available: http://chemteacher.chemeddl.org/services/chemteacher/index.php?option=com_content&view=article&id=66M