Infrared radiation

Infrared radiation (IR) is a type of radiant energy, with longer wavelengths than the visible light humans can see, but shorter wavelengths than radio waves. Its range extends from fairly small wavelengths near the color red, 700x10-9 m, to nearly a millimeter, 3x10-4 m.[2]
Effect of IR
Even though infrared radiation cannot be seen by the human eye, it can definitely be felt. Infrared energy is felt as heat because it interacts with molecules by exciting them, causing them to move faster which increases the internal temperature of the object absorbing the infrared energy. Although all wavelengths of radiant energy will heat surfaces that absorb them, infrared radiation is most common in daily life because of the "ordinary" objects that emit it as radiant heat (see blackbody radiation and Wien's Law for more information on this).[3] For example, humans at a temperature of 37°C[4] emit most of their radiant heat in the infrared range, as can be seen in Figure 1.
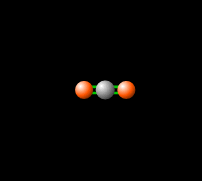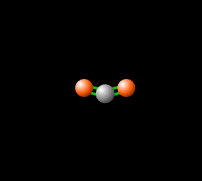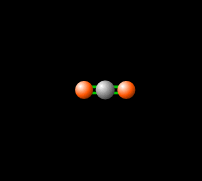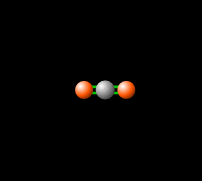
Around 50% of the Sun's energy to the Earth is in the form of infrared,[6] therefore the balance of this radiation in the atmosphere is crucial to keep a stable temperature and climate. Carbon dioxide in the atmosphere produces a greenhouse effect, because CO2 is able to absorb and re-emit infrared radiation as seen in Figure 2, unlike the gasses that make up most of the atmosphere (molecular oxygen, O2 about 21% and nitrogen, N2, about 78%).[7] This greenhouse effect is necessary for the livable temperatures on Earth, however an increasing level of greenhouse gases is contributing to an unstable warming of the Earth which is a cause for great concern. Read more about this imbalance here.
Since the infrared spectrum is of lower energy than visible light, this limits the amount of solar energy that can be harnessed with standard photovoltaic cells.
Use of IR

Infrared radiation has many applications, some being:
- Heating (cooking, saunas, industrial)
- Night vision (goggles, cameras)[9]
- Imaging (biological, mineral, defense, astronomy)
- Climatology and meteorology
For Further Reading
References
- ↑ Wikimedia Commons [Online], Available: http://upload.wikimedia.org/wikipedia/commons/0/0c/Infrared_dog.jpg
- ↑ CRISP, Electromagnetic waves [Online], Available: http://www.crisp.nus.edu.sg/~research/tutorial/em.htm
- ↑ Hyperphysics, Heat Radiation [Online], Available: http://hyperphysics.phy-astr.gsu.edu/hbase/thermo/stefan.html#c2
- ↑ R. A. Hinrichs and M. Kleinbach, "Heat and Work," in Energy: Its Use and the Environment, 4th ed. Toronto, Ont. Canada: Thomson Brooks/Cole, 2006, ch.4, sec.E, pp.111-114
- ↑ PhET Simulations, Molecules and Light [Online], Available: https://phet.colorado.edu/en/simulation/molecules-and-light
- ↑ Passive heating and cooling manual, Introduction to Solar Energy [Document], Available: http://www.azsolarcenter.com/design/documents/passive.DOC
- ↑ UCAR, Carbon Dioxide Absorbs and Re-emits Infrared Radiation [Online], Available: http://scied.ucar.edu/carbon-dioxide-absorbs-and-re-emits-infrared-radiation
- ↑ Wikimedia Commons [Online], Available: http://upload.wikimedia.org/wikipedia/commons/5/52/Nightvision.jpg
- ↑ American Technologies Network Corporation, How Night Vision Works [Online], Available: http://www.atncorp.com/HowNightVisionWorks

