Nuclear fusion
Nuclear fusion is a type of nuclear reaction where two light nuclei collide together to form a single, heavier nucleus. Fusion results in a release of energy because the mass of the new nucleus is less than the sum of the original masses. Based on the principle of mass-energy equivalence, this mass difference means that some mass that was "lost" has been converted into energy.[2] For elements lighter than iron, fusion often releases energy. For elements heavier than iron, it takes energy to cause fusion to happen. In order to make elements heavier than iron either a high energy particle beam is required, or a supernova.
Although the fusion of small atoms gives off a lot of energy, initiating this process requires a significant amount of energy. This energy is needed to overcome the Coulomb repulsion that exists between the protons the two different nuclei. Hydrogen atoms must be pushed close enough together so that the strong nuclear force can overcome the coulomb repulsion. The initial energy needed is a major factor which makes fusion difficult to achieve.[2]
Types of Fusion Reactions
There are several different types of fusion reactions, but most involve two isotopes of hydrogen known as deuterium and tritium. Some fusion reactions include:[3]
- Proton-proton chain: This type of fusion reaction is the one that takes place in the Sun. Two pairs of protons (two pairs of hydrogen atoms) collide and become two atoms of deuterium. Each deuterium them combines again with a proton (hydrogen) to form helium-3, which combine again and eventually form helium-4.
- Deuterium-deuterium reactions: a pair of deuterium atoms combine to form helium-3 and a neutron.
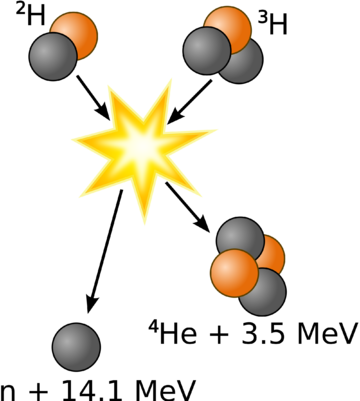
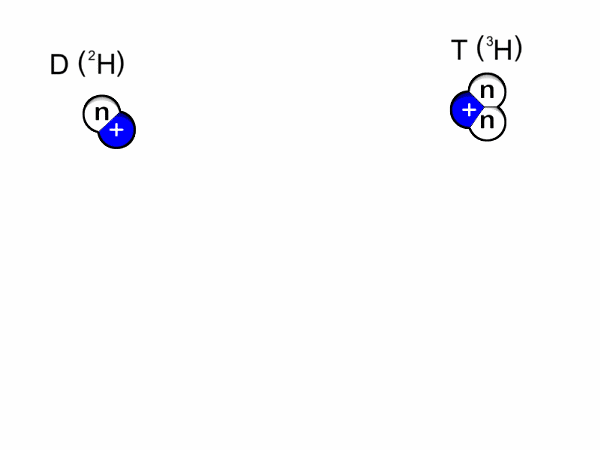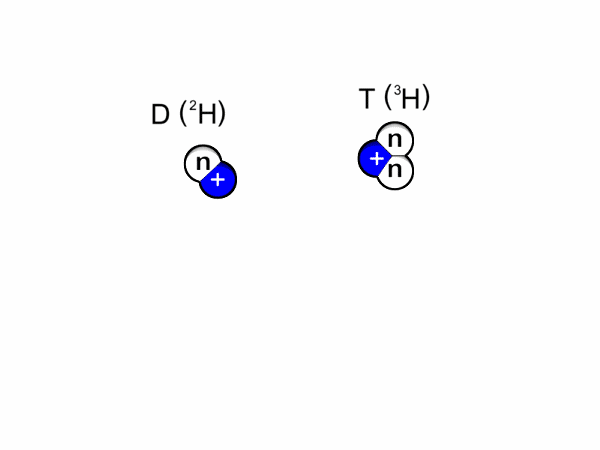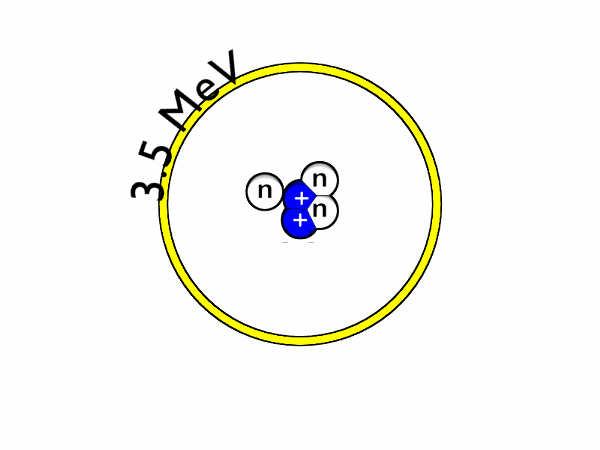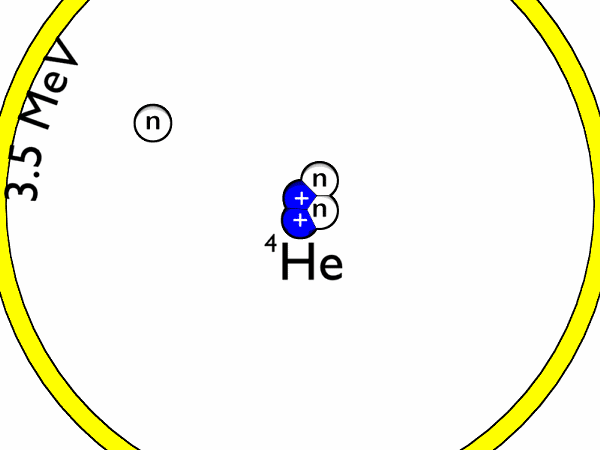
- Deuterium-trituim reactions: for this to occur, one atom of deuterium combines with one atom of tritium to form helium-4 and a neutron. Most of the energy released here is in the form of a high-energy neutron. This process is shown in Figure 1 and an animation is shown in Figure 2.
Use in Energy Generation
Currently, there are no large-scale fusion reactor that could provide energy for commercial use. This is because it has been difficult for scientists to create a controllable, non-destructive way of harnessing the energy released during fusion.[3] The process of fusion is difficult to control largely because of the extreme conditions necessary for the reactions to take place.
First, fusion requires both extremely high temperatures to give hydrogen atoms enough energy to overcome repulsion between the protons. Energy from microwaves or lasers must be used to heat hydrogen atoms to the necessary temperatures. At these temperatures, hydrogen is a plasma, and this plasma must be sufficiently contained for fusion to continue, and safety.[3] Second, high pressures are needed to squeeze hydrogen atoms close enough to fuse. This process is done by using intense magnetic fields, lasers, or ion beams.[3]
For potential nuclear energy sources, the deuterium-tritium fusion reaction is most likely because the conditions are less extreme.[2]
Currently, the largest fusion effort is the International Thermonuclear Experimental Reactor or ITER in France. This reactor began construction in 2013 and uses a confinement method known as a Tokamak. This Tokamak provides a way to magnetically confine the hot plasma required for fusion. The experimental phase of ITER is expected to begin in 2027.[5] For more information on this project, click here.
For Further Reading
References
- ↑ Wikimedia Commons. (August 4, 2015). Nuclear Fusion [Online]. Available: https://upload.wikimedia.org/wikipedia/commons/thumb/3/3b/Deuterium-tritium_fusion.svg/2000px-Deuterium-tritium_fusion.svg.png
- ↑ 2.0 2.1 2.2 HyperPhysics. (August 4, 2015). Nuclear Fusiom [Online]. Available: http://hyperphysics.phy-astr.gsu.edu/hbase/nucene/fusion.html
- ↑ 3.0 3.1 3.2 3.3 Craig Freudenrich. (August 4, 2015). Physics of Nuclear Fusion Reactions [Online]. Available: http://science.howstuffworks.com/fusion-reactor1.htm
- ↑ Wikimedia Commons. (July 31, 2015). Animated D-T Fusion [Online]. Available: https://commons.wikimedia.org/wiki/File:Animated_D-T_fusion.gif#/media/File:Animated_D-T_fusion.gif
- ↑ ITER. (August 4, 2015). ITER - the way to new energy [Online]. Available: https://www.iter.org/

