Temperature
Temperature is the reading obtained from a thermometer, which measures how hot or cold a substance is. At a microscopic level, temperature describes the average kinetic energy of molecules within a material or system. It is a measurable physical property of an object and can be considered with other measurable physical properties such as velocity, mass, and density (just to name a few).
Measurement
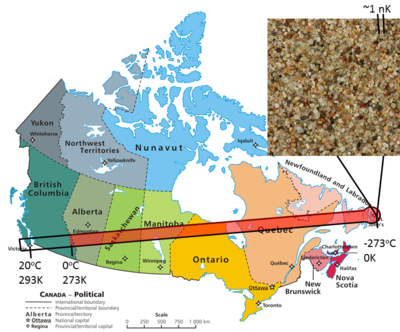
Temperature is typically seen in units of degrees Celsius or °C (in some countries the Fahrenheit scale is used); however, in the scientific community, temperature is seen in SI units—Kelvin or K (notice it's K not °K). Both Kelvin and Celsius scales have their advantages and disadvantages:
Celsius vs Kelvin
The Celsius and Kelvin scales increase by the same increments, meaning that to increase the temperature by 1°C results in the same increase of 1 K. The major difference is that when water freezes, a Celsius thermometer will read 0°C, and a Kelvin thermometer will read 273.15 K. The scales differ by 273.15. Thus, to convert from °C to K simply add 273; if a thermometer reads 31°C then the temperature in Kelvin is 304K.
Celsius is a much more "user friendly" or intuitive way to measure temperature; at 0°C—water freezes, and at 100°C—water boils. Celsius makes sense and it's much easier to judge how 25°C might feel than how 298 K would, and to be clear, 298 K would feel fine.
Why bother with Kelvin?
The Kelvin scale happens to be extremely useful (and necessary) when doing scientific calculations and measurements. Absolute zero is 0 K (converting to Celsius it's -273.15°C) and is the lowest temperature the laws of physics allow—there could never be temperatures below 0 K. In this respect it makes perfect sense to use the Kelvin scale, 0 being the "absolute" lowest and increasing from there. For more information (and a handy calculator) visit the page Kelvin.
Important misunderstanding
Because temperature is a measure of the microscopic energy of atoms (or molecules), the temperature doubles if the microscopic energy doubles. That being said, going from 10°C today to 20°C tomorrow isn't doubling the temperature (even though 20 is twice ten). A scientist would say it's 283 K, and here is where the problem arises: doubling 283 K is 566 K which converts to an extreme 293°C. Thankfully, 293°C would never happen on Earth, but this idea of a proportional change in temperature has led to some climate change confusion.
Climate change confusion
An overwhelming percentage of scientists agree that climate change is a major issue facing the world.[2] Most climate scientists predict a 1% increase in the average global temperature by the year 2100.[3] This number sounds negligible if thinking in Celsius, but a 1% change means the Kelvin scale must be used. Unfortunately, in order to do this calculation, the numbers have to be converted to Kelvin and after doing the proper conversions and calculations an increase by 1% on the Kelvin scale would actually result in an average global temperature of ~18°C by 2100 (because 291 K is roughly 1% warmer than the current average temperature of the planet, 288 K). This may not seem very high, but an increase of ~3°C is quite worrying.
For Further Reading
- Celsius
- Kelvin
- Fahrenheit
- Absolute zero
- Or explore a random page
References
- ↑ Modified from: By Siim Sepp (Own work) [CC BY-SA 3.0 (http://creativecommons.org/licenses/by-sa/3.0)], via Wikimedia Commons and By E Pluribus Anthony, transferred to Wikimedia Commons by Kaveh (log), optimized by Andrew pmk. (Own work) [Public domain], via Wikimedia Commons by Jason Donev January 12th, 2015.
- ↑ (2015, Feb. 6) http://www.ipcc.ch/publications_and_data/ar4/wg1/en/faq-8-1.html as one source on climate change consensus.
- ↑ (2015, Feb. 6) www.ipcc.ch/publications_and_data/ar4/wg1/en/ch10s10-es-1-mean-temperature.html the specific predictions about warming depend greatly on how much carbon dioxide human industry continues to produce.

