Smog

Smog is a specific type of air pollution. It is a combination of harmful pollutants (often appearing relatively low to the ground as a yellow-brown haze) that are introduced into the atmosphere by both natural and human induced processes.[2] It was first described over 5 decades ago as a mixture of smoke and fog, hence the name "smog"—but today it has a more specific definition and composition.
Smog is made up of many chemicals including nitrogen oxides (NOx), sulphur dioxide (SOx), carbon monoxide (CO), and volatile organic compounds (VOCs), but the two main components of smog are particulate matter (PM) and ground-level ozone (O3).[2][3]
Normal smog (often called London-type smog) is mainly a product of burning large amounts of high sulfur coal. Whereas photochemical smog is a more modern phenomena commonly produced by vehicle emissions in contact with sunlight—mostly from burning gasoline and diesel.[4] Photochemical smog forms in warm, densely populated cities with many vehicles. Visit the page here to learn more about photochemical smog.
This page does not include the chemical formulas in the formation of smog—to see them, along with more detail about smog, visit the UC Davis ChemWiki.
Formation
Smog forms when pollutants are released into the air. The pollutants are formed both naturally and by humans, however, the human-induced pollutants are of most concern due to the magnitude of pollutants produced by the burning and extraction of fossil fuels, which are known to cause extreme health effects. The location of smog formation is also of great concern, especially for human health, as a good portion of it is produced within cities where large portions of the population live.
One of the primary constituents, ozone, is created through chemical reactions between sunlight and certain pollutants. The other primary constituent, particulate matter, can also form through chemical reactions but is introduced to the atmosphere through other means as well. Wind may disperse these particles among land, thereby decreasing the amount within a given area. In addition, rainfall may wash these pollutants out of the local atmosphere,[2] however, this can result in other unwanted events like acid rain. When smog encounters an inversion layer (caused by warm areas in the upper atmosphere) it can stay over a region for an extended period of time—exposing people to its effects for longer. Visit the inversion layer page to see how this phenomena occurs.

Health Effects of Smog
The health effects from smog and its components can be severe, and depend on many variables. Smog is harmful when inhaled, with the severity of it depending on the amount inhaled, the types of pollutants contained in it, as well as the individual's age, weight, activity level, and well-being.[2][6] However studies show that any exposure to these pollutants is harmful, with extended exposure and higher doses obviously causing the most damage.
Types of effects smog has include:[2]
- Eye, nose and throat irritation
- Decreased lung function
- Aggravation of respiratory or heart disease
- In some cases, death
This effects can be seen in Figure 3 below, with more serious and rare effects at the top, and the more common yet milder effects towards the bottom.
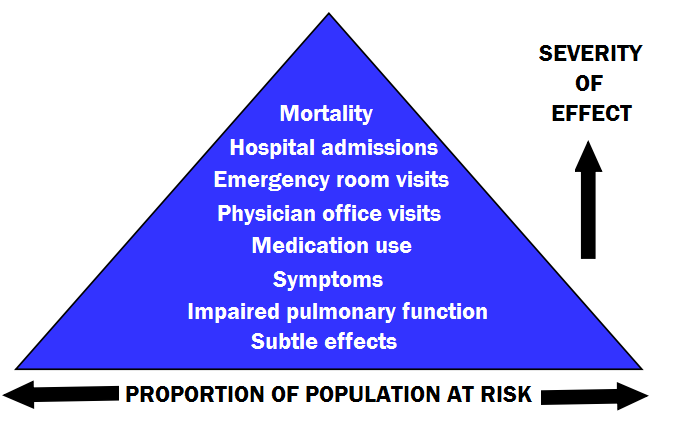
Specific health effects of pollutants
- Visit the main article of each to learn more about each pollutant
- Particulate matter (PM): Made up of solid and liquid particles that vary in size and composition, most PM gets caught in the nose and throat, and will not reach the lungs.[2] However, smaller particulates can make it to the lungs and are referred to as "respirable PM". These small particles can penetrate deep and cause damage. The cardio-respiratory (heart-lung) system are damaged the most from PM, resulting in infections, asthma, bronchitis, emphysema, black lung and more.
- Ozone (O3): An intense irritant and dangerous health threat, ozone can damage the eyes and nose, along with the throat and lungs. It is responsible for respiratory problems such as coughing and wheezing, and can reduce lung function.[2] Although irritated cells are replaced a few days after exposure, the replacement is not perfect and constant exposure may lead to aging of the lung and long-term effects.
- Nitrogen oxides (NOx): Responsible both directly and indirectly for health effects, NOx irritates lungs and lowers resistance to infection. Even short-term exposure can cause symptoms of coughing and shortness of breath. Indirectly it is involved in the formation of O3 and PM.[2]
- Sulfur dioxide (SOx): SO2 leads to eye irritation, shortness of breath and impaired lung functioning. It can penetrate deeply into the lungs, and when combined with water converts to sulfuric acid which the body is sensitive to. It is also one of the main contributors to the formation of small PM.
- Carbon monoxide (CO): CO is an air pollutant that effects health and in high concentrations is very deadly. It binds with haemoglobin in the blood, reducing its ability to carry oxygen and thereby starves vital tissues of oxygen. People with weaker immune systems and previous health problems are affected the most by high CO levels.[2]
Visit the Government of Canada for information of smog in Canada, and how to reduce your risk of its adverse effects.
Environmental Impacts
Smog affects much more than just human lives; it also has a large impact on the surrounding physical environment. Ozone and PM in particular cause damaging effects:[2]
Ozone can damage plant cells and inhibit their growth, reducing the amount of carbon dioxide that they take in during photosynthesis. This results in reduced agricultural yields in all sorts of crops. It can also affect synthetic materials like rubber, cotton and other materials, causing deterioration and even disintegration.
Particulate matter smothers plants limiting their exposure to the sun and inhibiting their ability to take in carbon dioxide, therefore, decreasing its photosynthesis capabilities. The chemical composition of the PM may also have an effect on the plants and their surrounding soil. Particulate matter causes the presence of haze in the air, thus reducing the clarity and colour of what can be seen. Humidity of the surrounding air also plays a role in its hazy effect. According to the U.S. Environmental Protection Agency, visibility is reduced from 144 kilometers to around 30 kilometers in certain parts of the United States.[2]
Visit Environment Canada for more details.
For Further Reading
- Pollution
- Pollutants
- Photochemical smog
- Particulate matter
- Ozone
- Air quality index
- Or explore a random page
References
- ↑ Wikimedia Commons [Online], Available: https://upload.wikimedia.org/wikipedia/commons/9/96/SmogNY.jpg
- ↑ 2.00 2.01 2.02 2.03 2.04 2.05 2.06 2.07 2.08 2.09 2.10 Prepared for Pollution Probe by Olivia Nugent. 2002. The Smog Primer.
- ↑ Environment Canada. (July 7, 2015). Smog - Air [Online], Available: https://www.ec.gc.ca/air/default.asp?lang=En&n=13D0EDAA-1
- ↑ UC Davis ChemWiki. (Accessed July 28, 2015). Smog [Online], Available: http://chemwiki.ucdavis.edu/Physical_Chemistry/Kinetics/Case_Studies%3A_Kinetics/Smog
- ↑ Wikimedia Commons [Online], Available: https://upload.wikimedia.org/wikipedia/commons/a/a4/Beijing_smog_comparison_August_2005.png
- ↑ Government of Canada. (July 7, 2015). Smog and your health [Online], Available: http://healthycanadians.gc.ca/healthy-living-vie-saine/environment-environnement/air/smog-eng.php
- ↑ Adapted from The Smog Primer (See Reference 1). Source: Stieb. DM; Pengelly LD, Arron N, Taylor SM, Raizenne M. Health effects of air pollution in Canada: Expert findings for the Canadian Smog Advisory Program. Can Respir J 1995;2(3):155-60.

