Atmosphere
An atmosphere is the layer of gases surrounding a planet. Many planets (and a number of moons) have atmospheres; some are thicker than Earth's (like Venus) and some are thinner (like Mars). Mercury and the Earth's moon have almost no atmosphere at all. Jupiter and the other gas giants have atmospheres that are remarkably thick and extensive.
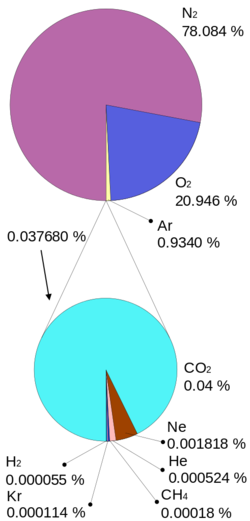
As seen in figure 1, the Earth's atmosphere is made mostly of nitrogen (78%) and oxygen (21%), with trace amounts of argon, carbon dioxide, and other gases. While not included here, water vapour can make up 0-4% of the Earth's atmosphere (and is not part of the listed percent in figure 1). The concentration of water vapour varies due to location and the time of day.[3] For a given temperature this amount is called relative humidity.
The atmosphere protects the surface of the Earth from harmful cosmic rays and holds the oxygen and carbon dioxide needed to support life.[4] The chemistry of the Earth's atmosphere is governed by chemical reactions, many of which come from life. Life is notably responsible for the atmospheric oxygen; without photosynthesis, the molecular oxygen in the atmosphere would chemically vanish.
In addition to being important for life, the atmosphere also allows the harvesting of a number of primary energy sources. For example, the oxygen in the atmosphere makes combustion of hydrocarbons (like those found in fossil fuels or biofuels) possible. The atmosphere also serves as a heat sink for certain types of heat engines (for example, a car engine couldn't run without dumping waste heat into the atmosphere; neither can certain types of power plants). More obviously, the wind is the atmosphere moving, so atmosphere is needed for wind power.
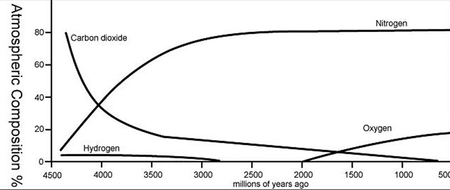
The atmosphere is responsible for the greenhouse effect, which makes the Earth warm enough to live on. In fact, most aspects of the Earth's weather and climate are an important consequence of the the atmosphere (the ocean being the other largest contributor). The chemistry of the atmosphere has changed over time. For example, for over a billion years there was almost no atmospheric oxygen. Figure 2 shows how the atmosphere was almost entirely carbon dioxide billions of years ago.
Because the atmosphere is a fluid, it moves quite readily around the surface of the Earth. This makes the atmosphere one of the primary vectors for pollution. The atmosphere is one of the places where humans put a fair amount of waste, especially waste from power plants. Some of this waste (like waste heat) is generally harmless, but most of it becomes atmospheric pollution (recall that pollution is anything that humans create that harms the environment, see pollution vs waste). Carbon dioxide waste (mostly from burning fossil fuels) is increasing the greenhouse effect in the atmosphere, leading to global warming and other forms of climate change.
For Further Reading
- Gas
- Primary energy
- Greenhouse effect
- Climate vs weather
- Or explore a random page
References
- ↑ (2014, Jan. 6). Atmosphere gas proportions.svg [Online]. Available: http://commons.wikimedia.org/wiki/File:Atmosphere_gas_proportions.svg
- ↑ Modified from figure 17.1: P. Bimblecombe and T. D. Davies, The Cambridge Encyclopedia of Earth Sciences, Cambridge, England: Cambridge University Press, 1981, p. 276.
- ↑ "Composition of the Atmosphere | North Carolina Climate Office", Climate.ncsu.edu, 2018. [Online]. Available: https://climate.ncsu.edu/edu/Composition. [Accessed: 07- Jun- 2018]
- ↑ "Atmosphere Protects", Forces.si.edu, 2018. [Online]. Available: http://forces.si.edu/atmosphere/02_04_00.html. [Accessed: 07- Jun- 2018].

