Secondary pollutant

Secondary pollutants are pollutants which form in the atmosphere. These pollutants are not emitted directly from a source (like vehicles or power plants). Instead, they form as a result of the pollutants emitted from these sources reacting with molecules in the atmosphere. Pollutants that are emitted into the environment from a source are called primary pollutants.
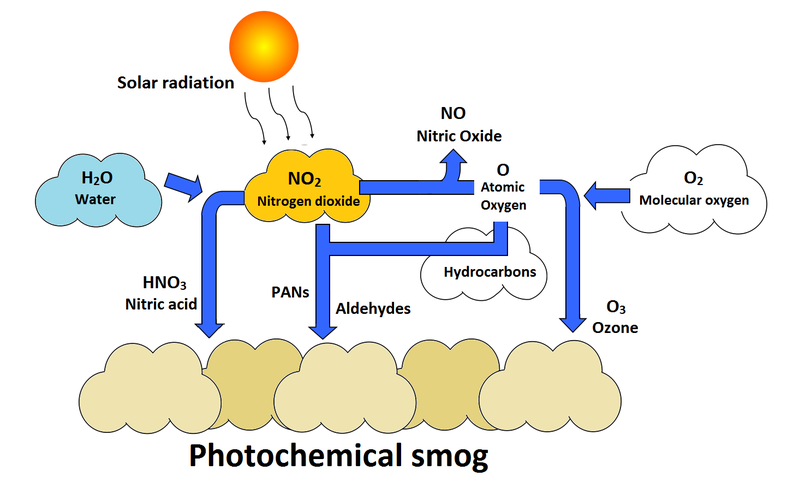
Secondary pollutants are concerning as they can be formed from many different compounds. The phenomena of photochemical smog (seen in high density cities, see Figure 1) is a result of the interactions of primary pollutants with other molecules in the air such as molecular oxygen, water and hydrocarbons. These combine to form yellow clouds that are harmful to humans. Photochemical smog is made up of various secondary pollutants like ozone, peroxyacyl nitrates (PANs), and nitric acid (seen in Figure 2).
Different types of secondary pollutants include:[2]
- Ozone (O3)
- Sulfuric acid and nitric acid (component of acid rain)
- Particulate matter
- Nitrogen dioxide (NO2)
- Peroxyacyl nitrates (PANs)
- and more
These substances essentially "cook up" in the atmosphere, and are typically found downwind of primary emissions due to the time it takes to produce them.[3] When primary pollutants cannot be dispersed due to inversion layers in the atmosphere, smog is formed over the area where they were produced. This is why smog is so prominent in warm, dense cities. Secondary pollutants are very sensitive to weather patterns.
For Further Reading
- Pollutant
- Pollution
- Primary pollutant
- Pollution vs waste
- Or explore a random page
References
- ↑ Wikimedia Commons [Online], Available: https://upload.wikimedia.org/wikipedia/commons/d/d1/Shanghaiairpollutionsunset.jpg
- ↑ LSC Atmospheric Sciences. (Accessed July 23, 2015). Secondary Pollutants [Online], Available: http://apollo.lsc.vsc.edu/classes/met130/notes/chapter18/secondary.html
- ↑ Tracey Holloway. Associate professor, Nelson Institute for Environmental Studies. University of Wisconsin-Madison. See: http://experts.news.wisc.edu/experts/65
- ↑ Adapted from Living In The Environment: G. Tyler Miller, Jr. and D. Hackett, "Photochemical and Industrial Smog," in Living in the Environment, 2nd ed. USA: Nelson , 2011, ch.20, sec.3, pp.465-471.

