Ideal gas approximation
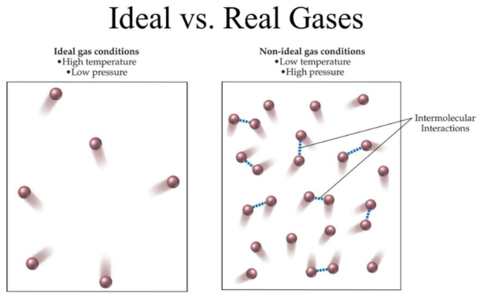
The ideal gas law allows us to determine what will happen to a contained system with an ideal gas inside, based on the equation:
There are three main approximations that must be made in order to use the ideal gas law:[2][3]
1. All collisions between atoms or molecules are assumed to be perfectly elastic in which there are no intermolecular attractive forces.
2. All gas particles are in constant motion and collisions between the gas molecules and the walls of the container cause the pressure of the gas.
3. The particles are so small that their volume is negligible compared with the volume occupied by the gas.
Density, Pressure and Temperature
Real gases approximate ideal gas behavior at relatively low density, low pressure, and high temperature.
At high temperatures, the gas molecules have enough kinetic energy to overcome intermolecular forces, but at low temperatures, the gas has less kinetic energy and thus the intermolecular forces are more prominent.[4] This makes assumptions #1 and #2 above more accurate.
At low gas densities and low pressures, there is lots of space between molecules, but at high densities the molecules are crowded and thus the volume of the gas particles is no longer negligible.[4] Lower pressures means fewer interactions, and those interactions can be more easily thought of as elastic collisions which makes assumption #2 more true. Fewer molecules in a volume makes assumption #3 more true.
Condensation Assumption
One final assumption for the ideal gas law is that an ideal gas "never condenses" regardless of changes in pressure, volume, and temperature. In reality, gases condense at high pressures or low temperatures. Condensation occurs when gas molecules can no longer overcome the intermolecular attractive forces due to a loss in kinetic energy.[5] For example, water vapour will condense to a liquid when the temperature is decreased. However, the ideal gas law assumes that the gas will not condense at high pressures or low temperatures.
For Further Reading
- Ideal gas law
- Kinetic energy
- Temperature
- Pressure
- Or explore a random page
References
- ↑ "Ideal Gases and Real Gases", Villanova College Chemistry Blog, 2017. [Online]. Available: https://villanovachemistry.wordpress.com/2017/06/01/ideal-gases-and-real-gases/. [Accessed: 06-May-2020].
- ↑ "Ideal Gas Law", Hyperphysics, 2020. [Online]. Available: http://hyperphysics.phy-astr.gsu.edu/hbase/Kinetic/idegas.html#c1. [Accessed: 28- Oct- 2020].
- ↑ "Ideal Gases and the Ideal Gas Law", Chemguide, 2020. [Online]. Available: https://www.chemguide.co.uk/physical/kt/idealgases.html. [Accessed: 28- Oct- 2020].
- ↑ 4.0 4.1 "Non-Ideal Gas Behavior", Chemistry LibreTexts, 2020. [Online]. Available: https://chem.libretexts.org/@go/page/38206.
- ↑ "Non-ideal (Real) Gases", Chemistry LibreTexts, 2020. [Online]. Available: https://chem.libretexts.org/@go/page/19180.

