Thermal mass
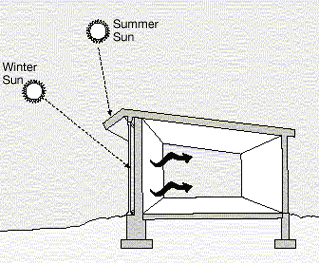
Thermal mass is a material property that can help reduce temperature fluctuations throughout the day. Thermal mass materials achieve this effect by absorbing heat during periods of high solar insolation, and releasing heat when the surrounding air begins to cool. When incorporated into passive solar heating and cooling technologies, thermal mass can play a significant role in reducing a building's energy use. As such, it is a property that helps reduce heating and cooling demand of buildings.
Properties of thermal mass
An ideal material for thermal mass will have:
- high heat capacity
- high material density
Heat capacity is the amount of heat energy required to change the temperature of an object by one degree Kelvin. The SI unit for heat capacity is Joule per Kelvin (J/K). The total amount of energy stored by a thermal mass system is proportional to the size of the system or material, therefore specific heat capacity (J/kg K), heat capacity per unit mass, and volumetric heat capacity (J/m3K), heat capacity per unit of volume, are common metrics used to determine a good thermal mass material.
Thermal mass materials
Listed below is a table of common building materials, their heat capacity, density, and volumetric heat capacity. As mentioned earlier, an optimal thermal mass material should have a high volumetric heat capacity.
| Material | Heat Capacity (J/K) | Density (kg/m3) | Volumetric Heat Capacity (MJ/m3K) |
|---|---|---|---|
| Water | 4.18 | 1000 | 4.18 |
| Gypsum | 1.09 | 1602 | 1.746 |
| Air | 1.0035 | 1.204 | 0.0012 |
| Concrete | 0.88 | 2371 | 2.086 |
| Brick | 0.84 | 2301 | 2.018 |
| Limestone | 0.84 | 2611 | 2.193 |
| Granite | 0.79 | 2691 | 2.125 |
| Wood | 0.42 | 550 | 0.231 |
Water has very attractive thermal mass properties and can be an attractive material for passive solar design. However, potential issues with water leakage and damage detour it's widespread use as a thermal mass storage medium. Concrete and brick have relatively high volumetric heat capacities and are common building materials. When used correctly with a solar wall or trombe wall, heating and cooling energy consumption can be greatly reduced.
Phase change materials
Traditional thermal mass materials use sensible heat to store and release passive energy from solar insolation. Phase change materials utilize latent heat storage and can absorb the same amount of solar energy in a much smaller volume.[3] As temperature increases, the material changes phases from solid to liquid. This is an endothermic reaction. When the surroundings cool (at night) the material changes from liquid to solid, an exothermic reaction. Stored heat is released into the building. The use of phase change materials is a relatively new concept in building science and there are many different materials being used for a wide array of applications.
Thermal mass and climate
During warm weather, thermal mass can absorb heat gained from sunlight. This will make interior space more comfortable, and greatly reduce cooling demand and the cost of air conditioning. During the night, a building cools and the stored heat energy is released into the interior space, reducing the heating demand. Thermal mass is most beneficial in climates where there is a large fluctuation between daytime and nighttime ambient temperatures. In areas with high nighttime temperatures, thermal mass can still be utilized, but the building must be ventilated at night with the cooler night air to exhaust the stored heat energy.[4]
For Further Reading
- Thermal insulation
- Heat transfer
- Energy efficiency
- Thermal efficiency
- Thermal convection
- Or explore a random page
References
- ↑ Wikimedia Commons. (August 6, 2015). Trombe Wall [Online]. Available: https://upload.wikimedia.org/wikipedia/commons/3/3d/Illust_passive_solar_d2_319pxW.gif
- ↑ Build Green Canada. (August 28, 2015). An explanation of thermal mass [Online]. Available: http://www.buildgreen.ca/2008/09/an-explanation-of-thermal-mass/
- ↑ F. Kuznik, D. David, K. Johannes, and J.-J. Roux, “A review on phase change materials integrated in building walls,” Renew. Sustain. Energy Rev., vol. 15, no. 1, pp. 379–391, Jan. 2011.
- ↑ G. P. Henze, T. H. Le, A. R. Florita, and C. Felsmann, “Sensitivity Analysis of Optimal Building Thermal Mass Control,” J. Sol. Energy Eng., vol. 129, no. 4, p. 473, 2007. 129, no. 4, p. 473, 2007.

