Heptane: Difference between revisions
J.williams (talk | contribs) m (1 revision imported) |
No edit summary |
||
| Line 1: | Line 1: | ||
[[Category:Done 2015-06 | [[Category:Done 2015-09-06]] | ||
<onlyinclude>'''Heptane''' is a [[hydrocarbon]] that can be burned as a [[fuel]]. | <onlyinclude>'''Heptane''' is a [[hydrocarbon]] that can be burned as a [[fuel]]. Its [[chemical]] formula is C<sub>7</sub>H<sub>16</sub></onlyinclude>, and it is a [[volatile]], colourless [[liquid]] that is odourless when pure.<ref name=pubchem> National Center for Biotechnology Information. (2015). ''Properties of Heptane'' [Online]. Available: http://pubchem.ncbi.nlm.nih.gov/compound/heptane [February 16,2015]. </ref> | ||
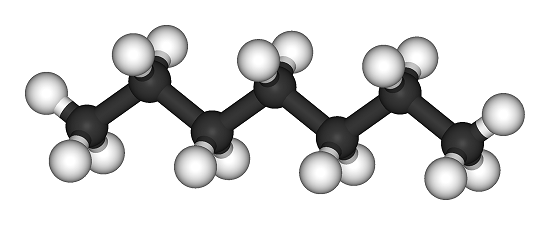
[[File:Heptane-3D-balls.png|400px|framed|center|Figure 1. Ball and stick model of heptane, the white is [[hydrogen]] and the black is [[carbon]].<ref>"Heptane-3D-balls". Licensed under Public Domain via Wikimedia Commons - http://commons.wikimedia.org/wiki/File:Heptane-3D-balls.png#mediaviewer/File:Heptane-3D-balls.png</ref>]] | [[File:Heptane-3D-balls.png|400px|framed|center|Figure 1. Ball and stick model of heptane, the white is [[hydrogen]] and the black is [[carbon]].<ref>"Heptane-3D-balls". Licensed under Public Domain via Wikimedia Commons - http://commons.wikimedia.org/wiki/File:Heptane-3D-balls.png#mediaviewer/File:Heptane-3D-balls.png</ref>]] | ||
Heptane is an important hydrocarbon as it, along with pure octane, sets the extreme ends of the [[octane rating]] scale. Heptane is used to set the standard zero point, which means that it is a fuel that burns so poorly within an engine that it could never realistically be used. Since higher octane ratings are better, heptane is a terrible fuel choice for a car since it burns very explosively, causing [[octane rating #Pre-Ignition and Knocking|engine knocking]]. As well as setting the zero point for octane rating, heptane is frequently used as a laboratory solvent due to it's low reactivity with other molecules. Many substances that will not dissolve in water do dissolve in heptane.<ref> Education Portal. (2015). ''Heptane: Structure, Uses, & Formula'' [Online]. Available: http://education-portal.com/academy/lesson/heptane-structure-uses-formula.html [February 16, 2015]</ref> Heptane undergoes [[hydrocarbon combustion]], combining with [[oxygen]] to form [[carbon dioxide]]. The balanced chemical equation for the complete combustion of heptane is: | Heptane is an important hydrocarbon as it, along with pure [[octane]], sets the extreme ends of the [[octane rating]] scale. Heptane is used to set the standard zero point, which means that it is a fuel that burns so poorly within an [[engine]] that it could never realistically be used. Since higher octane ratings are better, heptane is a terrible fuel choice for a car since it burns very explosively, causing [[octane rating #Pre-Ignition and Knocking|engine knocking]]. As well as setting the zero point for octane rating, heptane is frequently used as a laboratory solvent due to it's low reactivity with other molecules. Many substances that will not dissolve in [[water]] do dissolve in heptane.<ref> Education Portal. (2015). ''Heptane: Structure, Uses, & Formula'' [Online]. Available: http://education-portal.com/academy/lesson/heptane-structure-uses-formula.html [February 16, 2015]</ref> Heptane undergoes [[hydrocarbon combustion]], combining with [[oxygen]] to form [[carbon dioxide]]. The balanced chemical equation for the complete combustion of heptane is: | ||
| Line 18: | Line 18: | ||
| [[Molar mass]] || 100.21 [[kilogram|gram]]s/[[mole]] | | [[Molar mass]] || 100.21 [[kilogram|gram]]s/[[mole]] | ||
|- | |- | ||
| Melting | | [[Melting point]] || -91<sup>o</sup>C<ref name =elmhurst>Charles E. Ophardt. (2003). ''Virtual Chembook - Hydrocarbon Boiling Points'' [Online]. Available: http://www.elmhurst.edu/~chm/vchembook/501hcboilingpts.html [February 16,2015].</ref> | ||
|- | |- | ||
| Boiling | | [[Boiling point]] || 98<sup>o</sup>C<ref name =elmhurst/> | ||
|} | |} | ||
| Line 26: | Line 26: | ||
{{reflist}} | {{reflist}} | ||
[[Category:Uploaded]] | [[Category:Uploaded]] | ||
[[category:Ian edit]] | |||
Revision as of 19:23, 17 September 2015
Heptane is a hydrocarbon that can be burned as a fuel. Its chemical formula is C7H16, and it is a volatile, colourless liquid that is odourless when pure.[1]
Heptane is an important hydrocarbon as it, along with pure octane, sets the extreme ends of the octane rating scale. Heptane is used to set the standard zero point, which means that it is a fuel that burns so poorly within an engine that it could never realistically be used. Since higher octane ratings are better, heptane is a terrible fuel choice for a car since it burns very explosively, causing engine knocking. As well as setting the zero point for octane rating, heptane is frequently used as a laboratory solvent due to it's low reactivity with other molecules. Many substances that will not dissolve in water do dissolve in heptane.[3] Heptane undergoes hydrocarbon combustion, combining with oxygen to form carbon dioxide. The balanced chemical equation for the complete combustion of heptane is:
Properties
Below is a table of some of the basic properties of heptane.
| Chemical formula | C7H16 |
| Molar mass | 100.21 grams/mole |
| Melting point | -91oC[4] |
| Boiling point | 98oC[4] |
References
- ↑ National Center for Biotechnology Information. (2015). Properties of Heptane [Online]. Available: http://pubchem.ncbi.nlm.nih.gov/compound/heptane [February 16,2015].
- ↑ "Heptane-3D-balls". Licensed under Public Domain via Wikimedia Commons - http://commons.wikimedia.org/wiki/File:Heptane-3D-balls.png#mediaviewer/File:Heptane-3D-balls.png
- ↑ Education Portal. (2015). Heptane: Structure, Uses, & Formula [Online]. Available: http://education-portal.com/academy/lesson/heptane-structure-uses-formula.html [February 16, 2015]
- ↑ 4.0 4.1 Charles E. Ophardt. (2003). Virtual Chembook - Hydrocarbon Boiling Points [Online]. Available: http://www.elmhurst.edu/~chm/vchembook/501hcboilingpts.html [February 16,2015].