Octane: Difference between revisions
J.williams (talk | contribs) m (1 revision imported) |
No edit summary |
||
| Line 4: | Line 4: | ||
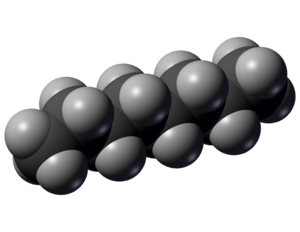
<onlyinclude>'''Octane''' is a [[hydrocarbon]] that can be burned as a [[fuel]]. It's chemical formula is C<sub>8</sub>H<sub>18</sub>, and it is a colourless liquid at room temperature with a characteristic "gasoline" odour.</onlyinclude><ref>National Center for Biotechnology Information. (2015). ''Properties of Octane'' [Online]. Available: http://pubchem.ncbi.nlm.nih.gov/compound/octane#section=Top [February 16, 2015].</ref> Its principle use is as a component of gasoline, and the performance of this gasoline depends on its [[octane rating]] (which gets its name from this [[molecule]], but is a little more complicated than just 'how much octane is there in the fuel). As is the case with other hydrocarbons, octane undergoes [[hydrocarbon combustion]], combining with [[oxygen]] to form [[carbon dioxide]]. The balanced chemical equation for the complete combustion of octane is: | <onlyinclude>'''Octane''' is a [[hydrocarbon]] that can be burned as a [[fuel]]. It's chemical formula is C<sub>8</sub>H<sub>18</sub>, and it is a colourless liquid at room temperature with a characteristic "gasoline" odour.</onlyinclude><ref>National Center for Biotechnology Information. (2015). ''Properties of Octane'' [Online]. Available: http://pubchem.ncbi.nlm.nih.gov/compound/octane#section=Top [February 16, 2015].</ref> Its principle use is as a component of gasoline, and the performance of this gasoline depends on its [[octane rating]] (which gets its name from this [[molecule]], but is a little more complicated than just 'how much octane is there in the fuel). As is the case with other hydrocarbons, octane undergoes [[hydrocarbon combustion]], combining with [[oxygen]] to form [[carbon dioxide]]. The balanced chemical equation for the complete combustion of octane is: | ||
< | <chem>2C_8H_{18} + 25(O_2)\leftrightarrow 16(CO_2) + 18(H_2O)</chem> | ||
Revision as of 22:15, 24 February 2019
Octane is a hydrocarbon that can be burned as a fuel. It's chemical formula is C8H18, and it is a colourless liquid at room temperature with a characteristic "gasoline" odour.[2] Its principle use is as a component of gasoline, and the performance of this gasoline depends on its octane rating (which gets its name from this molecule, but is a little more complicated than just 'how much octane is there in the fuel). As is the case with other hydrocarbons, octane undergoes hydrocarbon combustion, combining with oxygen to form carbon dioxide. The balanced chemical equation for the complete combustion of octane is:
Properties
Below is a table of some of the basic properties of octane.
| Formula | C8H18 |
| Molar mass | 114.23 grams/mole |
| Energy density | 47.9 MJ/kg [3] |
| Melting Point | -57oC[4] |
| Boiling Point | 125oC[4] |
References
- ↑ "N-octane-spaceFilling" Licensed under Public Domain via Wikimedia Commons - http://commons.wikimedia.org/wiki/File:N-octane-spaceFilling.png#mediaviewer/File:N-octane-spaceFilling.png
- ↑ National Center for Biotechnology Information. (2015). Properties of Octane [Online]. Available: http://pubchem.ncbi.nlm.nih.gov/compound/octane#section=Top [February 16, 2015].
- ↑ Glenn Elert. (2015). The Physics Hypertextbook - Chemical Potential Energy [Online]. Available: http://physics.info/energy-chemical/ [February 16, 2015].
- ↑ 4.0 4.1 Charles E. Ophardt. (2003). Virtual Chembook - Hydrocarbon Boiling Points [Online]. Available: http://www.elmhurst.edu/~chm/vchembook/501hcboilingpts.html [February 16,2015].