Ammonia
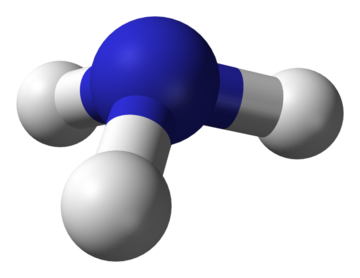
Ammonia is a chemical with the formula NH3. At room temperature, it is a colourless gas, with a pungent odour that is highly irritating. Ammonia is classified as a base, and is fairly corrosive. When put under enough pressure, ammonia gas forms a clear liquid and is generally shipped in this form. It is not highly flammable.[2] Figure 1 shows the molecular structure of ammonia. Some additional properties are shown in the table below.
| Chemical formula | NH3 |
| Molar mass | 17.031 grams/mole |
| Melting point | -77.73oC[3] |
| Boiling point | -33.34oC[3] |
Ammonia is a chemical that is essential for plant, animal, and human life. It is a natural component of the air, water, and soil and acts as a nitrogen source for plants and animals. However, in high quantities ammonia can be harmful for human health.[4]
Uses
Ammonia has a wide range of uses, but the majority of ammonia produced (~80%)[2] is used in agriculture as a fertilizer. This process allows soils to become more nitrogen rich. Ammonia can also be used as a refrigerant gas and is used in the manufacturing of plastics, explosives, textiles, pesticides, dyes, and other chemicals. Additionally, ammonia can be used as a cleaning solution in both household and industrial applications.[2]
In the petroleum industry, ammonia is used to neutralize acidic components of crude oil. In mining, ammonia is used to extract copper, nickel, and molybdenum from their ores. In waste water treatment, ammonia is used to control the pH of the water. Finally, ammonia is used to clean exhaust gases from smokestacks, removing sulfur oxides and NOx. This enhances the efficiency of electrostatic precipitators.[5]
Health Effects
Depending on how a person is exposed to ammonia, the health effects vary. When inhaled, ammonia acts as an irritant and burns the nose, throat, and respiratory tract. This inhalation of the corrosive chemical can result in respiratory distress. At lower concentrations this can lead to coughing. This exposure is more harmful to people with reduced respiratory functions.[2]
If ammonia comes into contact with the eyes or skin, it can cause irritation and burning. Extended exposure to ammonia can result in blindness and eye damage. If ingested, ammonia can burn the mouth, throat, and stomach.[2]
For Further Reading
- Hydrogen
- Nitrogen
- Base
- Or explore a random page
References
- ↑ Wikimedia Commons. (September 8, 2015). Ammonia 3D Model [Online]. Available: https://upload.wikimedia.org/wikipedia/commons/thumb/0/05/Ammonia-3D-balls-A.png/995px-Ammonia-3D-balls-A.png
- ↑ 2.0 2.1 2.2 2.3 2.4 Department of Health, New York. (September 8, 2015). The Facts About Ammonia [Online]. Available: https://www.health.ny.gov/environmental/emergency/chemical_terrorism/ammonia_tech.htm
- ↑ 3.0 3.1 PubChem. (September 8, 2015). Ammonia [Online]. Available: http://pubchem.ncbi.nlm.nih.gov/compound/ammonia
- ↑ The Fertilizer Institute. (September 8, 2015). Health Effects of Ammonia [Online]. Available: https://www.tfi.org/sites/default/files/documents/healthammonia2014.pdf
- ↑ RM Tech. (September 8, 2015). Uses of Ammonia [Online]. Available: http://www.rmtech.net/uses_of_ammonia.htm