Battery

A battery is a device that stores energy and then discharges it by converting chemical energy into electricity. Typical batteries most often produce electricity by chemical means through the use of one or more electrochemical cells.[2] Many different materials can and have been used in batteries, but the common battery types are alkaline, lithium-ion, lithium-polymer, and nickel-metal hydride. Batteries can be connected to each other in a series circuit or a parallel circuit.
There is a wide variety of batteries that are available for purchase, and these different types of batteries are used in different devices. Large batteries are used to start cars, while much smaller batteries can power hearing aids. Overall, batteries are extremely important in everyday life.
Cells
A cell is a single unit that produces electricity through some method. Generally speaking, cells generate power through a thermal, chemical or optical process.
A typical cell has two terminals (referred to as electrodes) immersed in a chemical (referred to as the electrolyte). The two electrodes are separated by a porous wall or bridge which allows electric charge to pass from one side to the other through the electrolyte. The anode—the negative terminal—gains electrons while the cathode—the positive terminal—loses electrons. This exchange of electrons allows a difference in potential or voltage difference to be developed between the two terminals—allowing electricity to flow.[2]
There can be a vast number of cells in a battery, from a single cell in an AA battery, to more than 7,100 cells in the 85 kWh Tesla Model S battery.[3]
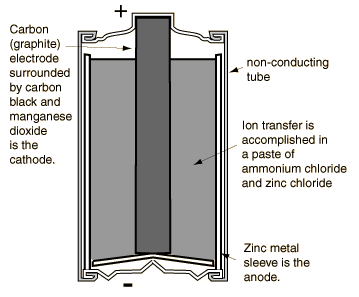
Primary cells ("dry")
In these cells a chemical action between the electrodes and electrolyte causes a permanent change, meaning they are not rechargeable.[2] These batteries are single use, which results in more waste from the use of these batteries since they are disposed of after a relatively short period of time.
Secondary cells ("wet")
This type of cell (referred to as wet due to using a liquid electrolyte) generates a current through a secondary cell in the opposite direction of the first/normal cell. This causes the chemical action to go in reverse, effectively being restored, meaning that they are rechargeable.[2] These batteries can be more expensive to purchase but generate less waste as they can be used several times.
Battery Capacity
Batteries are often rated in terms of their output voltage and capacity. The capacity is how long a particular battery will last in Ah (Ampere hours)[2]:
Batteries can also be rated by their energy capacity. This is either done in watt-hours or kilowatt-hours.
Phet Simulation
The University of Colorado has graciously allowed us to use the following Phet simulation. This simulation explores how batteries work in an electric circuit:
For Further Reading
For further information please see the related pages below:
- Series circuit
- Parallel circuit
- Direct current
- Dispatchable source of electricity
- Electrical grid
- Electric generator
- Or explore a random page!
References
- ↑ Wikimedia Commons [Online], Available: https://commons.wikimedia.org/wiki/File:Duracell_9_Volt_0849.jpg#/media/File:Duracell_9_Volt_0849.jpg
- ↑ 2.0 2.1 2.2 2.3 2.4 R.T. Paynter, “Basic Electric Components and Meters,” in Introduction to Electricity, 1rst ed. NJ: Prentice-Hall, 2011, ch. 3, sec. 3.4, pp. 89-94.
- ↑ Technology Metals Research. (Accessed July 28, 2015). Going Natural: The Solution To Tesla’s Graphite Problem [Online], Available: http://www.techmetalsresearch.com/2014/03/going-natural-the-solution-to-teslas-graphite-problem/
- ↑ Hyperphysics. (Accessed July 28, 2015). Carbon-zinc batteries [Online], Available: http://hyperphysics.phy-astr.gsu.edu/hbase/electric/battery.html

