Energy
The universe is made of matter (all ‘stuff’ in solid, liquid, or gas forms) and energy. Energy is the ability to create change. Careful scientific studies over centuries have found natural laws that govern energy—and these laws seem to be true everywhere in the known universe. As the US Department of Energy (US DOE) says:[2]
- Energy is a physical quantity that follows precise natural laws.
These include the law of conservation of energy, the first law of thermodynamics and the second law of thermodynamics.
Energy exists in how objects interact with each other. Often energy that can only be indirectly observed - by observing the processes that happen within a system. On Earth, everything from the weather to volcanoes comes from energy flowing through the various systems around us, as the US DOE goes on to say:[2]
- Physical processes on Earth are the result of energy flow through the Earth system.
Everything that physically happens, from light reflecting off of a rock to currents deep in the ocean happen because energy is flowing. Biological systems require energy too. In the words of the US DOE: [2]
- Biological processes depend on energy flow through the Earth system.
Every process in every living organism, including people, is driven by energy like photosynthesis and eating food. Everything from reproducing tiny cells to running down the street involves the transfer of energy.
Energy is often useful when it is transformed or transferred. This is often referred to as 'using energy', which is confusing because energy can't be used up—it just gets converted to a different form of energy! The two simplest ways of transferring energy are work and heat; both of which have specific definitions in the context of energy science. These definitions are slightly different from how the words are used in everyday language.
Part of what’s confusing about energy is that it is expressed using a lot of different units. These include units that are useful at the molecular level like electron volts (eV) to words used in the kitchen like calories. Physics classes tend to use units like joules, although chemists often favour calories. To make the units even more confusing the energy sector uses units like kilowatt-hours and BOE. There are also many large energy units useful for talking about energy use for whole countries like terawatt-hours and quads.
The rate (energy per unit of time) that energy is transferred (or 'used') is known as power. When energy is transferred quickly that means a large amount of power, when it's transferred slowly, it's less power. Visit energy vs power for a clear distinction.
Energy serves many uses
Energy provides many useful energy services that allow for a high quality of life that people living in modernized societies have grown accustomed to.[4] These services require a constant supply of primary fuels and primary flows to harness the needed energy. Specific services include powering vehicles (internal combustion engines), feeding populations, and generating electricity (see figure 2) for billions of people.
Energy also has the ability to cause damage or harm. For instance, burns are caused by an excess of thermal energy. Car accidents do a great deal of damage when the kinetic energy associated with driving down the street dissipates in a collision. Additionally, harnessing energy often leads to pollution and other environmental consequences like climate change.
Types of energy
Energy cannot be created or destroyed: this is known as the law of conservation of energy—meaning that energy must be harvested from some source. No process can create energy, no matter how nice that would be. However, many processes can transform energy from one type (like those found in nature) into another (like those useful for energy services). There are many different forms or types of energy that can be sorted into three main categories:
| Type of energy | Examples | Some ways of harnessing this energy |
|---|---|---|
| Kinetic energy | Macroscopic kinetic energy Microscopic kinetic energy Sound |
Wind power (macroscopic KE) Tidal power (macroscopic KE) Geothermal energy (microscopic KE) |
| Potential energy | Gravitational Chemical Nuclear Electric Elastic |
Hydroelectricity Fossil fuels Nuclear power Tidal power |
| Radiant energy | Electromagnetic (light) | Solar power |
For more information on how energy can't be created or destroyed but can only be taken from available resources in nature, please visit the page: law of conservation of energy
Energy conservation: where do we get the energy we use?
Since energy cannot be created (or destroyed) people extract energy from high density sources of energy called primary fuels, or physical processes that move energy called primary flows. The energy extracted from nature is called primary energy (the sum of all of our fuels and flows). Most primary energy comes from fuels like hydrocarbons and nuclear fuels. The rest of our primary energy is from flows like wind power and hydropower (see fuel vs flow). Energy can be found in nature in the form of fuels and flows, which can be harnessed and transformed into forms that are readily usable by humans. For example, the process of evaporation and rainfall allows rivers to keep flowing. We use the energy from the flow of water to create electricity, which had ultimately come from the sun. The energy changed form a number of times, but no energy was created or destroyed, just transferred.
Work
Science uses very specific definitions for words, which are often different from everyday use. To a physicist, the word work means to push (exert a force) an object some distance—a process that (by definition) requires an input of energy. For example, a person does work on a refrigerator by pushing it a distance across the kitchen—they apply a force with their hands over the entire distance—to do so, energy must be expended by the person! This energy gets transferred into the sound of a refrigerator scraping along the floor, and even a slight increase in temperature of the bottom of the fridge and floor.
For more information visit the page: work
Heat
Heat can be both a desired and undesired result of energy utilization. For example, thermal energy heats houses and meals, which is obviously desirable - especially in colder climates. However, heat is also a byproduct of friction, which isn't always sought after. For instance, intricate parts in vehicles use lubricating oil to reduce friction, but if the oil runs out those intricate parts will be exposed to high levels of friction which typically leads to high heating and serious damage within the engine.
For more information visit the page: heat
Light
Ultimately, most of the earth's energy comes from the nuclear fusion that takes place within the sun. This energy takes the form of light, which is otherwise known as electromagnetic radiation. In light, energy is transferred in little “packets” called photons that contain discrete amounts of energy that can propagate (as waves) over very long distances. When the photons hit an object, they transfer their energy to the object. It's important to note that all light, not just visible light, is like this. Light can have many different forms that are invisible to the unaided human eye. These different forms are classified by their wavelength and occupy a wide spectrum (known as the electromagnetic spectrum); differing ranges within this spectrum have differing levels of energy. From the highest energy to lowest are: gamma rays, x-rays, ultraviolet (UV), visible, infrared, microwave, and radio.
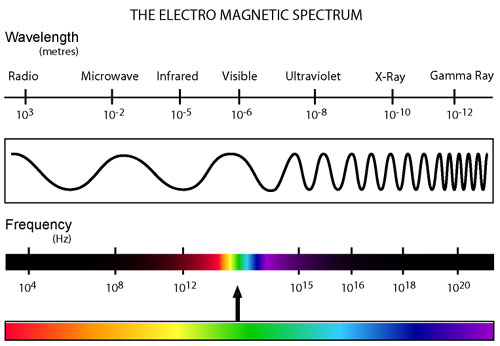
For more information visit the page: light
For Further Reading
- Law of conservation of energy
- Fuel vs flow
- Primary energy
- Energy conversion technology
- Or explore a random page
References
- ↑ Quintano, Anthony. (2014, Aug. 20). Lightning over New York City July 2013 [Online]. Available: https://www.flickr.com/photos/quintanomedia/9334838960/
- ↑ 2.0 2.1 2.2 ”Energy Literacy, Essential Principles and Fundamental Concepts for Energy Education” Online: https://www.energy.gov/sites/prod/files/2017/07/f35/Energy_Literacy.pdf Accessed: August 15th, 2017.
- ↑ By Nasa.gov http://www.nasa.gov/topics/earth/earthday/earthday_gallery.html [Public domain], via Wikimedia Commons accessed August 15th, 2017.
- ↑ Scott, Smelling Land, 1st edition. Vancouver, Canada: Canadian Hydrogen Association, 2007
- ↑ (2014, Aug. 20). Manufacturing Electronic Microwave Components. [Online] Available: http://www.thomasnet.com/articles/automation-electronics/electronic-microwave-manufacturing


