Gaseous diffusion uranium enrichment
Uranium found in the Earth's crust is made up of 99.289% 238U and 0.711% 235U, which are two isotopes of uranium. In order to use uranium in nuclear power plants, uranium ore must be mined, milled and then enriched to a higher percentage of 235U. Gaseous diffusion was the first economic enrichment process of uranium to be successfully developed.[1]

Methodology

Gaseous diffusion, like all uranium enrichment processes, utilizes the difference in mass between the 235U isotope and the 238U isotope. Because 238U possesses three more neutrons in its nucleus compared to 235U, it has a higher mass. The gaseous diffusion process is therefore based on the fact that, in a mixture of two gases, on average the lighter molecules travel faster than the heavier ones. [1] Due to this observation, if the gases are contained within a container, the lighter molecules will come into contact with the walls of the container more frequently. If the walls of the container were to be made of a semipermeable membrane, such that individual gas molecules could pass through but not enough to create a mass-flow through, then more of the lighter molecules will pass through the container. [1] The gas leaving the container is therefore slightly enriched in the lighter molecules and the gas that did not pass through the semipermeable membrane is depleted. Figure 2 shows a picture of a diffuser and its container, membrane and piping which is used in some gaseous diffusion plants.
Gaseous diffusion process
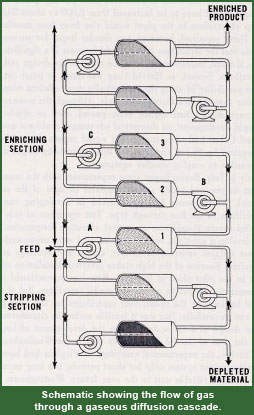
The gaseous diffusion process utilizes uranium hexafluoride, UF6, because although it is a solid at room temperature it is easily vaporized. [1] UF6 is not only convenient for its volatility, but also due to the fact that fluorine only consists of the isotope 19F, meaning the difference in molecular weights for UF6 are purely reliant on 235U and 238U.[1] Here arises another problem however, for the masses of the two uranium isotopes are so nearly equal there is very little separation of 235UF6 and 238UF6 with one pass through a diffuser.[1] Therefore a cascade process is needed to obtain any measurable amount of enrichment. Figure 3 on the right depicts diffusers set up in a cascade. The feed stream at diffuser 1 is the UF6 prior to enrichment (meaning it will contain 0.711% 235U and 99.289% 238U) and marks the start of the cascade. There will be hundreds to thousands of diffusers on the upward or enriching side as well as on the downward or depleted side. The slightly enriched UF6 is sent up the cascade process to the next diffuser where it will be enriched again. The slightly depleted UF6 will be sent downward through the cascade where it will also be enriched again. In this way, the enriched uranium keeps getting enriched and sent onward, and the depleted uranium also gets enriched and sent onward. The depleted uranium always gets sent downward where it will eventually be ejected from the downward stream as depleted uranium.
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 John R. Lamarsh, Anthony J. Baratta. (June 28, 2016). Introduction to Nuclear Engineering. Third Edition. Upper Saddle River, NJ, U.S.A:Prentice Hall, 2001.
- ↑ Energy.gov. (June 29, 2016). PORTSMOUTH SITE DESCRIPTION [Online]. Available: http://energy.gov/pppo/portsmouth-site-description
- ↑ Global Security (September 21, 2016). Gaseous Diffusion Uranium Enrichment [Online]. Available: http://www.globalsecurity.org/wmd/intro/u-gaseous.htm
- ↑ U.S. Department of Energy (September 21, 2016). The Manhattan Project [Online]. Available: https://www.osti.gov/opennet/manhattan-project-history/Events/1942-1944_ur/k-25_working.htm

