Isochore
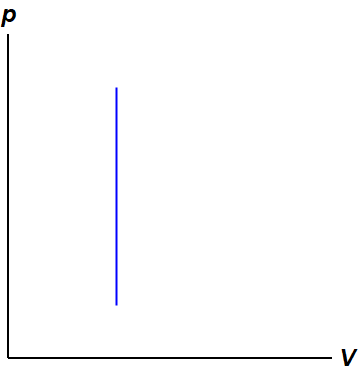
An isochoric process in the context of thermodynamics is a term used to describe a situation where the volume of a system remains constant. This process is modelled on a pressure volume diagram (PV diagram) as seen in Figure 1. For this process to be possible, the ideal gas must be in a rigid container that doesn't change in volume or let any molecules escape. Then, heat can be added or removed to change the pressure (and as a result also temperature) of the system.
For work to be done on the system, a force which moves it a nonzero distance must be applied. However, due to the system not expanding or compressing it is not 'moving any distance', so the work done during these processes will be zero.[2] This can be seen visually on the pressure-volume diagram (figure 1), as there is no area under the curve (since work is found by calculating the area under the curve).
One mechanism that uses the isochoric process is the expulsion of heat in heat engines that use diesel and gasoline. The waste heat is expelled right after the vehicle or machine receives power from combustion, but since the piston is not moving, the volume of the chamber is unchanging and the pressure drops. The process a gasoline engine undergoes is modelled by the Otto cycle (Figure 2) and diesel engines are modelled by the Diesel cycle (figure 3). An isochoric process is seen in the expulsion of heat in both process (4 to 1). Moreover, a gasoline engine exhibits an isochoric process during combustion as well (Figure 2, steps 2 to 3).
See the page on the Otto cycle and Diesel cycle for more information.
- Applications of the Isochore
- Figure 2. The PV diagram of the Otto Cycle[3]
- Figure 1. The PV diagram of the Diesel Cycle[4]
For Further Reading
- Pressure volume diagram
- Heat
- Ideal gas law
- Isobar
- Isothermal
- Adiabatic
- Or explore a random page
References
- ↑ Image made by Energyeducation team.
- ↑ R. Knight, Physics for scientists and engineers. Boston, Mass.: Addison-Wesley, 2012, p. 474
- ↑ Wikimedia Commons [Online], Available: https://en.wikipedia.org/wiki/Otto_cycle#/media/File:P-V_Otto_cycle.svg
- ↑ "File:Grafico p-v.JPG - Wikimedia Commons", Commons.wikimedia.org, 2018. [Online]. Available: https://commons.wikimedia.org/wiki/File:Grafico_p-v.JPG. [Accessed: 10- Jul- 2018].


![Figure 2. The PV diagram of the Otto Cycle[3]](/wiki/images/thumb/8/89/P-V_Otto_cycle.png/412px-P-V_Otto_cycle.png)
![Figure 1. The PV diagram of the Diesel Cycle[4]](/wiki/images/thumb/4/41/Diesel_cycle.jpg/402px-Diesel_cycle.jpg)