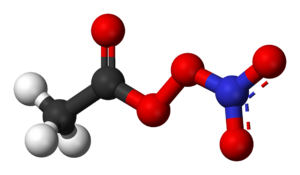
Peroxyacyl nitrate
Peroxyacyl nitrates or PANs, which are also referred to as acyl peroxy nitrates or APN, are a component of photochemical smog, produced in the atmosphere when oxidized volatile organic compounds combine with nitrogen dioxide ().[2] Figure 1 shows the structure of an example of PAN. PANs are a secondary pollutant since they form in the atmosphere after the emission of primary pollutants. Sources of the pollutants required to create PANs include motor vehicles, tobacco smoke, and the burning of fossil fuels.
Effects
PANs have many adverse effects in the human body such as reduced respiratory function (including emphysema and impaired breathing) and eye irritation. [3] Human exposure to PANs typically occurs in urban centers where automobile and industrial emissions are high.
PANs can remain in the atmosphere for about 3 months after formation, under cold conditions (-20°C and lower). However, in warmer areas, PANs persist only for a few hours. PANs that remain suspended in the atmosphere for extended periods of time are of concern, since they can be transported large distances by wind currents, thereby spreading their impact to other regions. Under these conditions, PANs can contribute to air pollution in places far away from their source.[4]
Decomposition of PANs can produce a variety of chemicals like carbon monoxide () and carbon dioxide ().[3]
For Further Reading
- Photochemical smog
- Volatile organic compound
- Secondary pollutant
- Pollutant
- Primary pollutant
- Or explore a random page
References
- ↑ Wikimedia Commons [Online], Available: https://en.wikipedia.org/wiki/Peroxyacetyl_nitrate#/media/File:Peroxyacetyl-nitrate-3D-balls.png
- ↑ LaFranchi, B. W.; Wolfe, G. M. (Accessed September 26, 2015). Closing the peroxy acetyl nitrate budget: observations of acyl peroxy nitrates (PAN, PPN, and MPAN) during BEARPEX 2007 [Online], Available: http://www.atmos-chem-phys.net/9/7623/2009/acp-9-7623-2009.html
- ↑ 3.0 3.1 HazMat. (Accessed September 26, 2015). Peroxyacetyl Nitrate (PAN) [Online], Available: http://www.hazmatmag.com/features/peroxyacetyl-nitrate-pan/
- ↑ F. Qureshi, University of Colorado Boulder. (Accessed September 26, 2015). Peroxyacetyl Nitrates: Ozone in the cooling PAN [Online], Available: http://atoc.colorado.edu/~toohey/PAN.pdf