Chemical isomer
Chemical isomers are molecules of the same chemical composition, but different molecular structure. In other words, two isomers will have the same number and type of atoms, but a different arrangement in space. Although these molecules are of the same composition, this different arrangement can result in a difference of physical and chemical properties (such as boiling point, melting point, etc).[1]
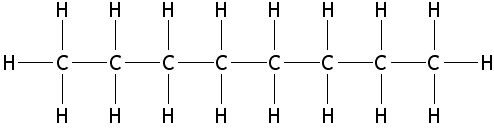
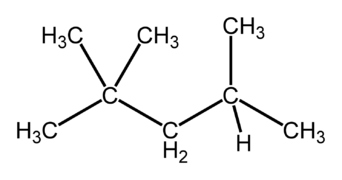
Isomers are very important for fuel choices when providing energy through combustion. Isomers of compounds burn differently, such as octane, which has 18 isomers.[2] For most applications the goal when burning a fuel is to have a stable burn, and certain isomers accomplish this better. For octane, the more linear isomers as visible in Figure 1 burn much quicker than isomers that are "branched" like in Figure 2. The branched structures allow the fuel to burn slowly and evenly, therefore they are of more desire.[2] Octane rating, which is the number visible on gas pumps, is a measure of how much iso-octane (Figure 2) is in a fuel mixture. The more iso-octane present in the mixture, the better the burn.
Figure 1. n-Octane, a more linear isomer of octane compared to its other forms.[3]
Figure 2. iso-octane, a more branched isomer of octane, used in most fuels and gives the octane rating for a fuel.[4]
Visit the UC Davis Chemwiki for a more in depth look at chemical isomers.
For Further Reading
- Hydrocarbon combustion
- Octane
- Octane rating
- Chemical
- Or explore a random page
References
- ↑ Compound Interest, A Bried Guide to Types of Isomerism in Organic Chemisty [Online], Available: http://www.compoundchem.com/2014/05/22/typesofisomerism/
- ↑ 2.0 2.1 eHow, Importance of Isomers in Fuels [Online], Available: http://www.ehow.com/about_5587676_importance-isomers-fuels.html
- ↑ Wikimedia Commons [Online], Available: http://commons.wikimedia.org/wiki/File:Octane-in-full.png
- ↑ Wikimedia Commons [Online], Available: http://upload.wikimedia.org/wikipedia/commons/7/79/Isooctane.png