Ionizing radiation
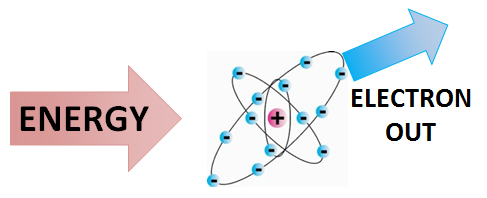
Ionizing radiation is a specific type of energy transmission (radiation) that has enough energy to remove electrons from atoms, turning atoms into ions. This means that when an atom or particle is hit by one of these energy carriers, the energy is not absorbed, but rather it breaks the bond between the electron and the nucleus of the atom, "ionizing" it. These resulting particles are therefore known as ions. Generally speaking, the incoming energies of alpha and beta decay particles, and gamma ray photons are higher than the ionization energies of atoms and molecules. This means that alpha, beta and gamma radiation are all examples of ionizing radiation.[2] Ionizing radiation is important as it can produce a number of physiological side-effects, both negative (such as cancer), and positive (such as cancer treatment).[3]
Geiger counters work by using this ionization. Geiger counters are instruments that detect ionizing radiation. They can't detect non-ionizing radiation as they rely on ions to make an electrical signal.
Uses of Ionizing Radiation
Different forms of ionizing radiation have different uses. Radiation therapy, a cancer treatment, relies on beta decay and uses its ionizing properties to kill cancer cells.[4] Alpha radiation also has use in the medical field, with targeted alpha therapy being used to kill cancer.
Ionizing radiation also has uses outside of the medical field. The ionizing properties of americium results in its use in smoke detectors. Inside the smoke detector alpha particles from the americium are released. This in turn ionizes the air inside the detector. Smoke in the detector absorbs this alpha radiation, so if smoke is present the ionization is altered and the alarm is triggered.[5] In addition, cobalt—a source of gamma ionizing radiation—is used to sterilize medical equipment and irradiate food, killing bacteria and pasteurizing the food.[6]
Health Effects
The biological effects of radiation vary depending on how much exposure a person has, the length of exposure they are expose to, and what type of radiation they are exposed to. Ionizing radiation is produced when radioactive materials decay, causing damage to living tissues that cannot always be repaired.[7] This exposure can be therapeutic, like treating cancer, or harmful. Chronic exposure to radiation can lead to cancer (as a result of damage at the cellular or molecular level). Exposure, especially in rapidly growing cells can cause mutations that can be harmful. Effects from acute exposure to ionizing radiation appear quickly, and include burns and radiation poisoning. The symptoms of radiation poisoning include nausea, weakness, hair loss, and diminished organ function and this radiation sickness can result in death if the dose is high enough.[7]
Knowing Nuclear
This video was created by the Energy Education team to further explain ionizing radiation
For Further Reading
- Radiation
- Energy
- Alpha decay
- Gamma decay
- Photon
- Or explore a random page
References
- ↑ Created internally by a member of the Energy Education team.
- ↑ R. Knight. (May 20, 2015). Physics for Scientists and Engineers, 3rd ed. U.S.A.: Pearson
- ↑ Hyperphysics. (May 19, 2015). Ionizing Radiation [Online]. Available: http://hyperphysics.phy-astr.gsu.edu/hbase/mod4.html
- ↑ ChemTeacher. (July 22, 2015). Beta Decay [Online]. Available: http://chemteacher.chemeddl.org/services/chemteacher/index.php?option=com_content&view=article&id=66M
- ↑ BBC Bitesized. (July 22, 2015). Uses of Radiation [Online]. Available: http://www.bbc.co.uk/schools/gcsebitesize/science/ocr_gateway_pre_2011/living_future/4_nuclear_radiation2.shtml
- ↑ US EPA. (May 14, 2015). Gamma Rays [Online]. Available: http://www.epa.gov/radiation/understand/gamma.html#use
- ↑ 7.0 7.1 US EPA. (July 8, 2015). Health Effects: Radiation [Online]. Available: http://www.epa.gov/radiation/understand/health_effects.html#q1

