Propane: Difference between revisions
J.williams (talk | contribs) m 1 revision imported |
No edit summary |
||
| Line 1: | Line 1: | ||
[[Category:Done | [[Category:Done 2018-06-15]] | ||
[[File:Propane | <translate> | ||
<!--T:1--> | |||
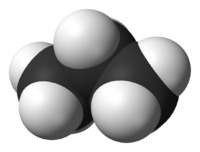
[[File:Propane-3D-vdW-B.png|thumbnail|right|200px|Figure 1. Space-filling model of a propane molecule. The black spheres represent carbon and the white spheres represent hydrogen<ref>Ben Mills. (2014, Dec. 12). ''Propane-3D-vdW-B'' [Online]. Available: http://commons.wikimedia.org/wiki/File:Propane-3D-vdW-B.png#mediaviewer/File:Propane-3D-vdW-B.png</ref>]] | |||
<onlyinclude>'''Propane''' is an [[alkane]] with the chemical formula C<sub>3</sub>H<sub>8</sub>. As a type of [[hydrocarbon]], it can undergo [[hydrocarbon combustion]], which gives off [[heat]]. Propane is one of the [[hydrocarbon]] components of raw [[natural gas]], which is a type of [[fossil fuel]].</onlyinclude><ref>“NATURAL GAS FAQs,” Pacific Northern Gas RSS. [Online]. Available: http://www.png.ca/natural-gas-faqs/. [Accessed: 24-May-2017]</ref> Propane is usually removed from natural gas before being shipped to customers, but is also sold separately as a [[fuel]] on its own. | |||
< | <!--T:2--> | ||
[[File:Propane | [[File:Propane tank.jpg|thumbnail|200px|Figure 2. A standard propane storage tank.<ref>''Personal photo submitted by a member of the Energy Education team.''</ref>]] | ||
Propane | <!--T:3--> | ||
==Properties== | Propane is normally in a [[gas]]eous state during consumption. However, it is normally stored as a [[liquefied propane gas]] in tanks such as the one shown in Figure 2. | ||
Propane has one of the highest [[energy density|energy densities]] (50.3 MJ/kg) of any hydrocarbon, second only to [[methane]].<ref name=chemical/> | |||
<!--T:4--> | |||
Propane is a hydrocarbon commonly used in domestic settings. It is normally seen in appliances such as [[barbecue]]s, patio [[heater]]s, and [[camping stove]]s. | |||
==Properties== <!--T:5--> | |||
<!--T:6--> | |||
Below is a table of some of the basic properties of propane. | Below is a table of some of the basic properties of propane. | ||
<!--T:7--> | |||
{| class="wikitable" | {| class="wikitable" | ||
|- | |- | ||
| Chemical formula || C<sub>3</sub>H<sub>8</sub> | | Chemical formula || C<sub>3</sub>H<sub>8</sub> | ||
|- | |- | ||
| | | Molecular Mass || 44.1 [[kilogram|gram]]s/[[mole]] | ||
|- | |- | ||
| Energy density || 50.3 MJ/kg<ref name=chemical>(2014, Jul. 1). ''Chemical Potential Energy'' [Online]. Available: http://physics.info/energy-chemical/</ref> | | Energy density || 50.3 MJ/kg<ref name=chemical>(2014, Jul. 1). ''Chemical Potential Energy'' [Online]. Available: http://physics.info/energy-chemical/</ref> | ||
| Line 21: | Line 34: | ||
|} | |} | ||
==Combustion | ==Combustion Reaction== <!--T:8--> | ||
Propane | |||
<html><iframe src=' | <!--T:9--> | ||
Propane releases its [[chemical energy]] by undergoing [[hydrocarbon combustion]]. Below is a [[hydrocarbon combustion]] animation showing the net reaction that occurs when propane combines with [[oxygen]]. | |||
<!--T:10--> | |||
<center> C<sub>3</sub>H<sub>8</sub> + 5O<sub>2</sub> → 3CO<sub>2</sub> + 4H<sub>2</sub>O + Heat Energy ([[Enthalpy]]) </center> | |||
<!--T:11--> | |||
<html><iframe src='https://energyeducation.ca/simulations/combustion/combustion_propane.html' width='900px' height='330px' style='border:none;position:relative;left:-35px'></iframe></html> | |||
<!--T:12--> | |||
The [[hydrocarbon combustion]] reaction releases heat [[energy]] and is an example of an [[exothermic reaction]]. The reaction also has a negative [[enthalpy]] change (ΔH) value. | |||
</translate> | |||
==For Further Reading== | |||
*[[Hydrocarbon]] | |||
*[[Hydrocarbon combustion]] | |||
*[[Chemical energy]] | |||
*[[Chemical bond]] | |||
*Or explore a [[Special:Random|random page]] | |||
==References== | ==References== | ||
<languages /> | |||
{{reflist}} | {{reflist}} | ||
[[Category:Uploaded]] | [[Category:Uploaded]] | ||
Revision as of 21:10, 24 June 2018
<translate>
Propane is an alkane with the chemical formula C3H8. As a type of hydrocarbon, it can undergo hydrocarbon combustion, which gives off heat. Propane is one of the hydrocarbon components of raw natural gas, which is a type of fossil fuel.[2] Propane is usually removed from natural gas before being shipped to customers, but is also sold separately as a fuel on its own.

Propane is normally in a gaseous state during consumption. However, it is normally stored as a liquefied propane gas in tanks such as the one shown in Figure 2. Propane has one of the highest energy densities (50.3 MJ/kg) of any hydrocarbon, second only to methane.[4]
Propane is a hydrocarbon commonly used in domestic settings. It is normally seen in appliances such as barbecues, patio heaters, and camping stoves.
Properties
Below is a table of some of the basic properties of propane.
| Chemical formula | C3H8 |
| Molecular Mass | 44.1 grams/mole |
| Energy density | 50.3 MJ/kg[4] |
| Melting Point | -190oC[5] |
| Boiling Point | -42oC[5] |
Combustion Reaction
Propane releases its chemical energy by undergoing hydrocarbon combustion. Below is a hydrocarbon combustion animation showing the net reaction that occurs when propane combines with oxygen.
The hydrocarbon combustion reaction releases heat energy and is an example of an exothermic reaction. The reaction also has a negative enthalpy change (ΔH) value.
</translate>
For Further Reading
References
<languages />
- ↑ Ben Mills. (2014, Dec. 12). Propane-3D-vdW-B [Online]. Available: http://commons.wikimedia.org/wiki/File:Propane-3D-vdW-B.png#mediaviewer/File:Propane-3D-vdW-B.png
- ↑ “NATURAL GAS FAQs,” Pacific Northern Gas RSS. [Online]. Available: http://www.png.ca/natural-gas-faqs/. [Accessed: 24-May-2017]
- ↑ Personal photo submitted by a member of the Energy Education team.
- ↑ 4.0 4.1 (2014, Jul. 1). Chemical Potential Energy [Online]. Available: http://physics.info/energy-chemical/
- ↑ 5.0 5.1 (2015, Jan. 29). Boiling Points and Structures of Hydrocarbons [Online]. Available: [http://www.elmhurst.edu/~chm/vchembook/501hcboilingpts.html

