Dopant
Doping is the practice of introducing very small amounts of certain foreign atoms into the crystal lattice of a semiconductor to modify its electrical properties. These foreign atoms are referred to as dopants. Often they add charge carriers to the semiconductor by creating either an excess or a deficiency of electrons around the foreign atom.[1] This leads to two distinct types of doping, p-type and n-type. P-type and n-type doping allows for the creation of important circuit components like diodes and transistors.[2]
N-type doping
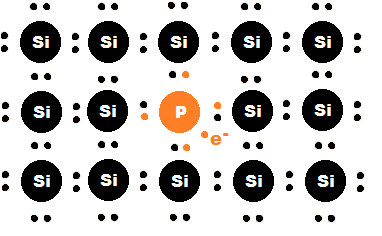
Adding atoms with a greater number of valence electrons than silicon to a silicon lattice results in the creation of free electrons. Since silicon has four valence electrons, the most common n-type dopants are atoms with five valence electrons like phosphorus, antimony, and arsenic. When these atoms are incorporated into the silicon lattice, four valence electrons are used to bond with the surrounding silicon atoms, leaving one valence electron free to move into the conduction band (see Figure 1).[4] N-type doping therefore dramatically increases the electrical conductivity of the semiconductor by increasing the number of free charge carriers.[5]
P-type doping
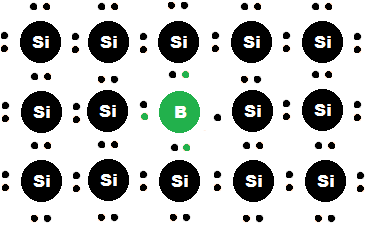
When an atom with one fewer valence electron than silicon is added to a silicon lattice, electron deficiencies are created (see Figure 2). These deficiencies are often called "holes".[6] These holes readily accept free electrons and complement n-type semiconductors as the excess electrons of the n-type can be absorbed by the p-type. This property is integral to the p-n junction, a vital component in the operation of a diode. P-type dopants have three valence electrons, and they include boron, aluminum, and gallium.[7]
For more information on doping semiconductors please see hyperphysics.
For Further Reading
For further information please see the related pages below:
- Conductor
- Metal
- Photovoltaic cell
- AC to DC adapter
- Or explore a random page!
References
- ↑ Doping of semiconductors [Online]. Available: http://www.mrl.ucsb.edu/~seshadri/old/MATRL100A/class12.pdf
- ↑ Semiconductors and Doping [Online]. Available:https://www.pa.msu.edu/courses/1997spring/PHY232/lectures/semiconductors/semiconductors.html
- ↑ 3.0 3.1 This image was created by part of the Energy Education team.
- ↑ Doping [Online]. Available:http://www.pveducation.org/pvcdrom/pn-junction/doping
- ↑ N-type Semiconductor [Online]. Available:http://hyperphysics.phy-astr.gsu.edu/hbase/solids/dope.html#c3
- ↑ P-type Semiconductor [Online]. Available:http://hyperphysics.phy-astr.gsu.edu/hbase/solids/dope.html#c4
- ↑ The Doping of Semiconductors [Online]. Available:http://hyperphysics.phy-astr.gsu.edu/hbase/solids/dope.html

