Layers of the atmosphere
Earth's atmosphere is a mixture of gases that cover the planet's surface and are held there by gravity. The atmosphere has different compositions, temperatures, and other properties at different heights. Notably, the gases in the lowest layers are under more pressure from the weight of the gases in the layers above them; for this reason, the atmosphere has a higher density at the planet's surface and becomes less and less dense, or "thinner," at higher altitudes.
The chemical composition, temperatures, movement and densities of the atmosphere at different heights differ widely enough that we recognize five distinct layers.[1] Their names, altitudes and qualities are as follows:
| Name and Image[2] | Temperature | Description |
|---|---|---|
 |
Temperatures are only relevant for individual molecules, which vary widely from near absolute zero (-273°C) at night or in the shade to thousands of degrees in direct sunlight. | Atmospheric temperature is essentially constant with altitude from this point upwards. Particles are still bound by Earth's gravity but no longer behave as a gas. Only the lightest gases are present: mainly Hydrogen, but also Helium, Carbon dioxide, and atomic Oxygen. There is no clear upper boundary to the exosphere, which is a transition area between the atmosphere and space. Many satellites orbit at these heights.[3] |
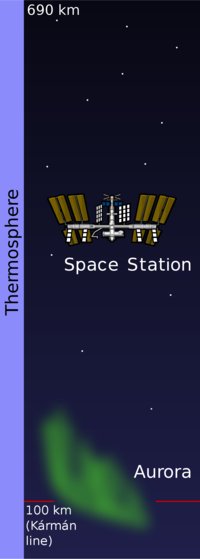 |
Temperatures increase with higher altitude, beginning around -120°C and reaching up to 2000°C at the upper limits.[1] | The mixture of gas molecules is no longer consistent and molecules with higher mass sink to lower layers. Intense solar UV radiation causes ionization of the gas molecules and warms them to extreme temperatures. However, the air here is incredibly thin (near vacuum) and would not feel hot to the touch since it is nearly empty. Some satellites and the international space station are located at these altitudes, and solar winds cause Aurora near the north and south poles.[3] The upper boundary is called the thermopause or exobase. |
 |
Temperatures range from -15°C to -120°C, decreasing with higher altitude[1] | The mesosphere is too high for aircraft and too low for spacecraft to maintain orbit. Most meteors burn up as they fall through this layer of the atmosphere.[3][1] It's upper boundary, the mesopause, is the coldest place on Earth.[3] |
 |
Averages range from -51°C to -15°C, increasing with higher altitude | Calm and stable with very few clouds, ideal for commercial jet flight. The stratosphere contains the ozone layer, which consists of a band of Ozone gas (O3). Ozone blocks and absorbs energy from incoming UVA and UVB radiation, which warms the stratospheric air.[3] This layer makes up 19% of the atmosphere by mass but contains almost no water vapour.[1] The stratopause marks the upper boundary and is the altitude where temperature reaches a maximum before decreasing in the mesosphere. |
 |
Averages range from 17°C to -51°C, generally decreasing with higher altitude[1] | The troposphere is heated mainly by the Earth's surface, and rising warm air causes rapid vertical mixing of the gases. Most weather phenomena occur here,[1] and the troposphere contains 99% of the atmosphere's water vapour.[3] Marking its upper boundary is the tropopause, an area where temperature reaches a minimum before increasing with altitude in the stratosphere. The gases in this layer represent 75% of the atmosphere by mass and are comprised mainly of Nitrogen (78%) and Oxygen (21%) (see atmospheric oxygen).[3] |
For Further Reading
- Atmospheric oxygen
- Atmospheric pressure
- Greenhouse effect
- Pollution
- Climate change
- Or explore a random page
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 National Oceanic and Atmosphere Administration. (2013, July 22). Layers of the Atmosphere [Online]. Available: http://www.srh.noaa.gov/jetstream/atmos/layers.htm [Accessed 14 October 2013].
- ↑ Kelvin Case [CC BY-SA 2.5 (http://creativecommons.org/licenses/by-sa/2.5)], via Wikimedia Commons, Atmosphere layers [Online]. Available: https://commons.wikimedia.org/wiki/File:Atmosphere_layers-en_af.png. Accessed 18 August 2013.
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 3.6 NASA Jet Propulsion Laboratory. (2013). Atmosphere layers [Online]. Available: http://airs.jpl.nasa.gov/maps/satellite_feed/atmosphere_layers/ [Accessed 14 October 2013].

