Energy loss

When energy is transformed from one form to another, or moved from one place to another, or from one system to another there is energy loss. This means that when energy is converted to a different form, some of the input energy is turned into a highly disordered form of energy, like heat. Functionally, turning all of the input energy into the output energy is nigh impossible, unless one is deliberately turning energy into heat (like in a heater). As well, whenever electrical energy is transported through power lines, the energy into the power lines is always more than the energy that comes out at the other end. Energy losses are what prevent processes from ever being 100% efficient.
Types of Energy Losses
Energy undergoes many conversions and takes on many different forms as it moves. Every conversion that it undergoes has some associated "loss" of energy. Although this energy doesn't actually disappear, some amount of the initial energy turns into forms that are not usable or we do not want to use.[2] Some examples of these losses include:
- Heat energy, potentially as a result of air drag or friction. Heat energy is the most easily dissipated form of energy.
- Light energy is frequently energy seen in combustion, and is a type of wave motion.
- Sound energy is another type of wave motion caused by the vibration of molecules in the air. Like heat energy, sound is a type of energy that is generally lost.
Overall, the goal is to reduce the amount of energy lost to increase efficiency. As well, collisions that are inelastic refer to collisions where there is some "loss" of energy during the collision.[3]
For more information on inelastic collisions, see HyperPhysics.
Energy Lost in Electricity Use
Electricity use is a good example that illustrates energy loss in a system. By the time the energy associated with electric power reaches the user, it has taken many forms. Initially, the process begins with the creation of the electricity through some method. For example, the burning of coal in a power plant takes the chemical energy stored in the coal and releases it through combustion, creating heat that produces steam. From here the steam moves turbines and the mechanical energy here turns a generator to produce electricity. A typical coal fired electrical plant is around 38% efficient,[2] so ~1/3 of the initial energy content of the fuel is transformed into a usable form of energy while the rest is lost. Further losses occur during the transport of this electricity. In the transmission and distribution of electricity in the United States, the EIA estimates that about 6% of the electricity is lost in these processes.[4] Finally, the electricity reaches its destination. This electricity could reach an incandescent light bulb wherein a thin wire is heated until it glows, with a significant amount of energy being lost as heat, shown in Figure 1. The resulting light contains only about 2% of the energy content of the coal used to produce it.[2] Changing to CFL light bulbs can improve this by about 4x, but that only takes it up to 8% of the initial chemical energy in the coal.
Fuels have enormous energy contents, but very little actually ends up as usable energy and most is lost. These energy losses result in extremely inefficient processes, some of these come from fundamental limitations like the second law of thermodynamics, but some provide opportunities for better engineering.
Energy Lost in Vehicles
There are also significant energy losses within a car's internal combustion engine. The chemical energy from the gasoline (or diesel) - which originates from the Sun as it is a fossil fuel - is then converted into heat energy, which presses on pistons in the engine. The mechanical energy is then transported to the wheels which increases the kinetic energy of the car. Some of this kinetic energy is lost to the sound of the engine, light from combustion, and to heat energy from the friction between the road and the tires. Current vehicles are only able to use around 20% of the energy content of the fuel as power, the rest is lost.[2] An example of these energy losses is shown in Figure 2. Although efficiencies can be improved, they can only be increased to a degree because of principles of thermodynamics.
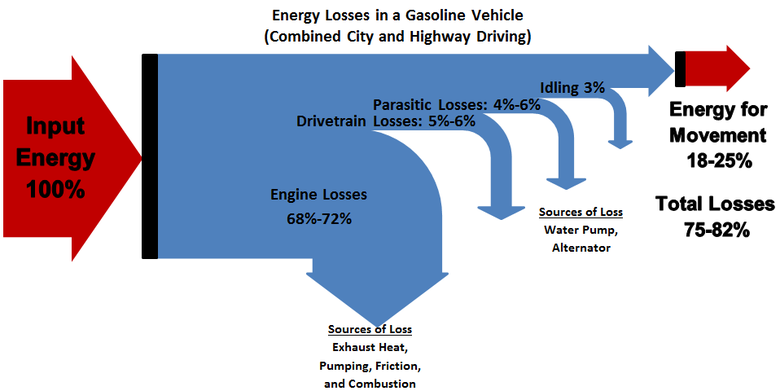
Idling, using accessories such as air conditioners, and aerodynamic drag can further increase losses in a vehicle.[6]
For Further Reading
- Law of conservation of energy
- Heat engine
- Heat pump
- Energy conversion technology
- Or explore a random page
References
- ↑ Wikimedia Commons. (May 27, 2015). Incandescent Light Bulb [Online]. Available: http://commons.wikimedia.org/wiki/File:Incandescent_light_bulb_on_db.jpg#/media/File:Incandescent_light_bulb_on_db.jpg
- ↑ 2.0 2.1 2.2 2.3 The National Academies. (May 27, 2015). Energy Sources[Online]. Available: http://www.nap.edu/reports/energy/sources.html
- ↑ HyperPhysics. (May 27, 2015). Inelastic Collisions [Online]. Available: http://hyperphysics.phy-astr.gsu.edu/hbase/inecol2.html
- ↑ EIA. (May 27, 2015). Electricity Losses [Online]. Available: http://www.eia.gov/tools/faqs/faq.cfm?id=105&t=3
- ↑ Created internally by a member of the Energy Education team. Adapted from: http://www.fueleconomy.gov/feg/atv.shtml
- ↑ Consumer Energy Center. (May 27, 2015). Energy Losses in a Vehicle [Online]. Available: http://www.consumerenergycenter.org/transportation/consumer_tips/vehicle_energy_losses.html

