Thermodynamic cycle
Thermodynamic cycle refers to any closed system that undergoes various changes due to temperature, pressure, and volume, however, its final and initial state are equal.[1] This cycle is important as it allows for the continuous process of a moving piston seen in heat engines and the expansion/compression of the working fluid in refrigerators, for example. Without the cyclical process, a car wouldn't be able to continuously move when fuel is added, or a refrigerator would not be able to stay cold.
Visually, any thermodynamic cycle will appear as a closed loop on a pressure volume diagram. The following are examples of thermodynamic cycles:
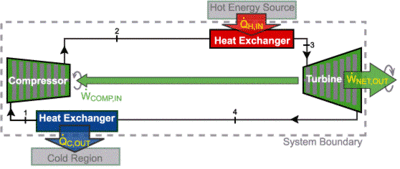
Brayton cycle
This a thermodynamic cycle used in some heat engines. Notably, it is used for gas turbine engines and some jet engines. The cycle consists of compressing ambient air, mixing the air with fuel, then igniting the mixture, which expands, doing work. In many Brayton cycle engines, the hot air can then be recycled, heating the fresh air coming through.
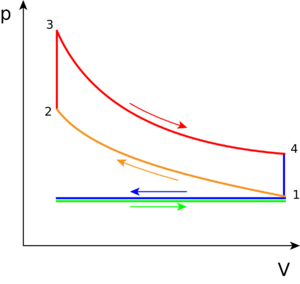
Otto Cycle
This cycle describes how heat engines turn gasoline into motion. Like other thermodynamic cycles, this cycle turns chemical energy into thermal energy and then into motion. The Otto cycle describes how internal combustion engines (that use gasoline) work, like automobiles and lawn mowers. Without the cycle, the vehicle would suddenly stop, and wouldn't be able to provide continuous motion.
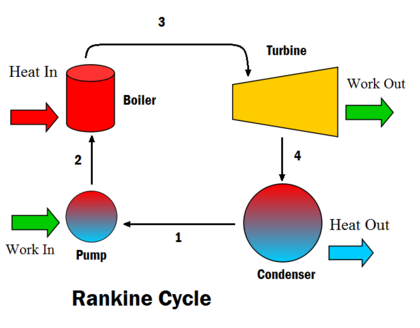
Rankine cycle
This process is widely used by power plants such as coal-fired power plants or nuclear reactors. In this mechanism, a fuel is used to produce heat within a boiler, converting water into steam which then expands through a turbine producing useful work. The schematic diagram shows there is no 'break' between the steps in a Rankine cycle, and exemplifies the process is a continuous loop.
For Further Reading
- Thermodynamics
- Pressure volume diagram
- System and surrounding
- Ideal gas law
- Or explore a random page
References
- ↑ A. Atkins and M. Escudier, A dictionary of mechanical engineering.
- ↑ Wiley.com, 2018. [Online]. Available: https://www.wiley.com/college/moran/CL_0471465704_S/user/tutorials/tutorial9/tut9n_content.html. [Accessed: 31- Jul- 2018].
- ↑ Wikimedia Commons [Online], Available: https://en.wikipedia.org/wiki/Otto_cycle#/media/File:P-V_Otto_cycle.svg
- ↑ Made internally be a member of the Energy Education team, adapted from Energy Systems Engineering by F. Vanek, L. Albright and L. Angenent