Nuclear fusion in the Sun: Difference between revisions
J.williams (talk | contribs) m (1 revision imported) |
No edit summary |
||
| Line 1: | Line 1: | ||
[[Category: | [[Category:Ashley edit]] | ||
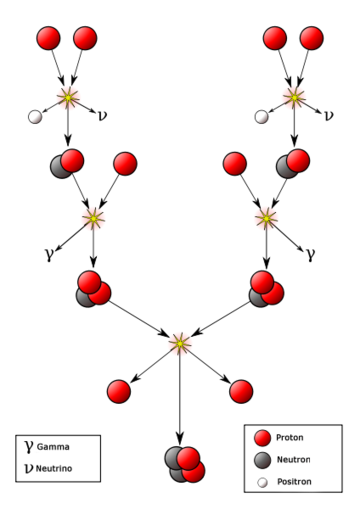
[[File:sunfusion2.png|360px|thumb|right|Figure 1. The proton-proton fusion process that is the source of energy from the Sun.<ref>Wikimedia Commons. (July 30, 2015). ''Fusion in the Sun'' [Online]. Available:https://upload.wikimedia.org/wikipedia/commons/0/03/Fusion_in_the_Sun_it.png</ref> ]] | [[File:sunfusion2.png|360px|thumb|right|Figure 1. The proton-proton fusion process that is the source of energy from the Sun.<ref>Wikimedia Commons. (July 30, 2015). ''Fusion in the Sun'' [Online]. Available:https://upload.wikimedia.org/wikipedia/commons/0/03/Fusion_in_the_Sun_it.png</ref> ]] | ||
| Line 11: | Line 11: | ||
# Two protons within the Sun fuse. Most of the time the pair breaks apart again, but sometimes one of the protons transforms into a [[neutron]] via the [[weak nuclear force]]. Along with the transformation into a neutron, a positron and neutrino are formed. This resulting proton-neutron pair that forms sometimes is known as [[deuterium]]. | # Two protons within the Sun fuse. Most of the time the pair breaks apart again, but sometimes one of the protons transforms into a [[neutron]] via the [[weak nuclear force]]. Along with the transformation into a neutron, a positron and neutrino are formed. This resulting proton-neutron pair that forms sometimes is known as [[deuterium]]. | ||
# A third proton collides with the formed deuterium. This collision results in the formation of a helium-3 nucleus and a [[gamma decay|gamma ray]]. These gamma rays work their way out from the core of the Sun and are released as [[sunlight]]. | # A third proton collides with the formed deuterium. This collision results in the formation of a helium-3 nucleus and a [[gamma decay|gamma ray]]. These gamma rays work their way out from the core of the Sun and are released as [[sunlight]]. | ||
# Two helium-3 nuclei collide, creating a helium-4 nucleus plus two extra | # Two helium-3 nuclei collide, creating a helium-4 nucleus plus two extra protons that escape as two hydrogen. Technically, a [[beryllium|beryllium-6]] nuclei forms first but is unstable and thus disintegrates into the helium-4 nucleus. | ||
The final helium-4 atom has less mass than the original 4 protons that came together (see [[E=mc2]]). Because of this, their combination results in an excess of energy being released in the form of heat and light that exits the Sun, given by the [[mass-energy equivalence]]. To exit the Sun, this energy must travel through many layers to the [[Sun#Layers of the Sun|photosphere]] before it can actually emerge into space as sunlight. Since this proton-proton chain happens frequently - 9.2 x 10<sup>37</sup> times per second - there is a significant release of energy.<ref name="RE2"/> Of all of the [[mass]] that undergoes this fusion process, only about 0.7% of it is turned into energy. Although this seems like a small amount of mass, this is equal to 4.26 million metric [[tonne]]s of matter being converted to energy per second.<ref name="RE2"/> Using the mass-energy equivalence, we find that this 4.26 million metric tonnes of matter is equal to about 3.8 x 10<sup>26</sup> [[joule]]s of energy released per second! | The final helium-4 atom has less mass than the original 4 protons that came together (see [[E=mc2]]). Because of this, their combination results in an excess of energy being released in the form of heat and light that exits the Sun, given by the [[mass-energy equivalence]]. To exit the Sun, this energy must travel through many layers to the [[Sun#Layers of the Sun|photosphere]] before it can actually emerge into space as sunlight. Since this proton-proton chain happens frequently - 9.2 x 10<sup>37</sup> times per second - there is a significant release of energy.<ref name="RE2"/> Of all of the [[mass]] that undergoes this fusion process, only about 0.7% of it is turned into energy. Although this seems like a small amount of mass, this is equal to 4.26 million metric [[tonne]]s of matter being converted to energy per second.<ref name="RE2"/> Using the mass-energy equivalence, we find that this 4.26 million metric tonnes of matter is equal to about 3.8 x 10<sup>26</sup> [[joule]]s of energy released per second! | ||
== | ==For Further Reading== | ||
For more information on the energy that comes from the Sun, see: | For more information on the energy that comes from the Sun, see: | ||
* [[Sun]] | * [[Sun]] | ||
* [[Solar radiation]] | * [[Solar radiation]] | ||
* [[Insolation]] | * [[Insolation]] | ||
* [[Solar energy to the Earth]] | * [[Solar energy to the Earth]] | ||
* [[Temperature of the Earth]] | * [[Temperature of the Earth]] | ||
* [[Solar power]] | * [[Solar power]] | ||
*Or explore a [[Special:Random|random page]] | |||
==References== | ==References== | ||
{{reflist}}[[Category:Uploaded]] | {{reflist}}[[Category:Uploaded]] | ||
Latest revision as of 03:17, 16 June 2020
The energy from the Sun - both heat and light energy - originates from a nuclear fusion process that is occurring inside the core of the Sun. The specific type of fusion that occurs inside of the Sun is known as proton-proton fusion.[2]
Inside the Sun, this process begins with protons (which is simply a lone hydrogen nucleus) and through a series of steps, these protons fuse together and are turned into helium. This fusion process occurs inside the core of the Sun, and the transformation results in a release of energy that keeps the sun hot. The resulting energy is radiated out from the core of the Sun and moves across the solar system.[3] It is important to note that the core is the only part of the Sun that produces any significant amount of heat through fusion (it contributes about 99%).[3] The rest of the Sun is heated by energy transferred outward from the core.
Steps
The overall process of proton-proton fusion within the Sun can be broken down into several simple steps. A visual representation of this process is shown in Figure 1. The steps are:[4]
- Two protons within the Sun fuse. Most of the time the pair breaks apart again, but sometimes one of the protons transforms into a neutron via the weak nuclear force. Along with the transformation into a neutron, a positron and neutrino are formed. This resulting proton-neutron pair that forms sometimes is known as deuterium.
- A third proton collides with the formed deuterium. This collision results in the formation of a helium-3 nucleus and a gamma ray. These gamma rays work their way out from the core of the Sun and are released as sunlight.
- Two helium-3 nuclei collide, creating a helium-4 nucleus plus two extra protons that escape as two hydrogen. Technically, a beryllium-6 nuclei forms first but is unstable and thus disintegrates into the helium-4 nucleus.
The final helium-4 atom has less mass than the original 4 protons that came together (see E=mc2). Because of this, their combination results in an excess of energy being released in the form of heat and light that exits the Sun, given by the mass-energy equivalence. To exit the Sun, this energy must travel through many layers to the photosphere before it can actually emerge into space as sunlight. Since this proton-proton chain happens frequently - 9.2 x 1037 times per second - there is a significant release of energy.[3] Of all of the mass that undergoes this fusion process, only about 0.7% of it is turned into energy. Although this seems like a small amount of mass, this is equal to 4.26 million metric tonnes of matter being converted to energy per second.[3] Using the mass-energy equivalence, we find that this 4.26 million metric tonnes of matter is equal to about 3.8 x 1026 joules of energy released per second!
For Further Reading
For more information on the energy that comes from the Sun, see:
- Sun
- Solar radiation
- Insolation
- Solar energy to the Earth
- Temperature of the Earth
- Solar power
- Or explore a random page
References
- ↑ Wikimedia Commons. (July 30, 2015). Fusion in the Sun [Online]. Available:https://upload.wikimedia.org/wikipedia/commons/0/03/Fusion_in_the_Sun_it.png
- ↑ Fraser Cain. (July 30, 2015). Fusion in the Sun [Online]. Available: http://www.universetoday.com/18707/fusion-in-the-sun/
- ↑ 3.0 3.1 3.2 3.3 Jerry Coffey. (July 31, 2015). How Does The Sun Produce Energy? [Online]. Available: http://www.universetoday.com/75803/how-does-the-sun-produce-energy/
- ↑ Karl Tate. (July 30, 2015). Proton Fusion, the Sun's Power Source, Explained [Online]. Available: http://www.space.com/26956-proton-fusion-sun-power-source-infographic.html

