Anthropogenic carbon emissions

Anthropogenic carbon emissions are the emissions of various forms carbon - the most concerning being carbon dioxide - associated with human activities. These activities include the burning of fossil fuels, deforestation, land use changes, livestock, fertilization, etc., that result in a net increase in emissions.[2]
Increasing Carbon Levels
A major concern is the emission of carbon dioxide (CO2) - a type of greenhouse gas - which contributes to global warming and ocean acidification. CO2 and other compounds of carbon are exchanged throughout carbon pools in the global carbon cycle. The natural carbon cycle is kept in a near perfect balance, however human emissions are input into the cycle which cause a net increase in concentrations of carbon in the atmosphere, soil, and oceans, as seen in Figure 1.[3]
Methane (CH4), carbon monoxide, and black carbon are all present in the atmosphere, and each have varying effects on the planet. The amount of each of these in the atmosphere has been increasing rapidly: the amount of carbon dioxide in the atmosphere has increased by about 40% since the industrial era, and methane by about 150%.[3] These levels had been relatively stable for around 10 thousand years prior to this.[4]
See carbon cycle for more information on increasing levels of carbon in Earth's crucial systems.
Measuring Emissions
Scientists have had to work hard to show that climate change is caused by the greenhouse gasses that humans emit. Part of the reason that this was tricky is that nature does emit nearly 20 times more CO2 into the atmosphere than humans do, but it's the new carbon that humans introduce that changes the amount of carbon in the system.[3] This section highlights techniques and observational evidence to support the impact that humans have had on the environment.
There are many scientific methods that allow for the measurements of human emissions, two of which will be talked about here:
- The first method is the investigation of carbon-14 (14C), a radioactive isotope of carbon. 14C is found naturally in atmospheric CO2, however it is not present in fossil fuels that humans extract from underground. This is because fossil fuels were formed millions of years ago, far longer than the half-life of 14C (t1/2 = 5730 years), meaning that all of the 14C has decayed away.[3] This means that carbon emitted by these fuels does not contain any 14C. By measuring levels of 14C and CO2 over time, it is found that the vast majority of excess CO2 does not contain 14C and therefore can be attributed to fossil fuels.
- The second method relates to changes in atmospheric oxygen levels. It is employed in order to determine whether this excess CO2 is indeed from human fossil fuels, or if it is instead from the likes of volcanic eruptions. This method works off of basic hydrocarbon combustion knowledge - when a fossil fuel is burned, its hydrocarbons consume oxygen in order to give off energy, along with water vapour and carbon dioxide. Therefore oxygen levels are expected to decrease when humans burn fossils fuels, and when measuring levels over time it is found that they do, the amounts of which are in accordance with measured carbon dioxide levels. If the emissions were instead dominantly from volcanoes, there would be no measured decrease in atmospheric oxygen levels.[3]
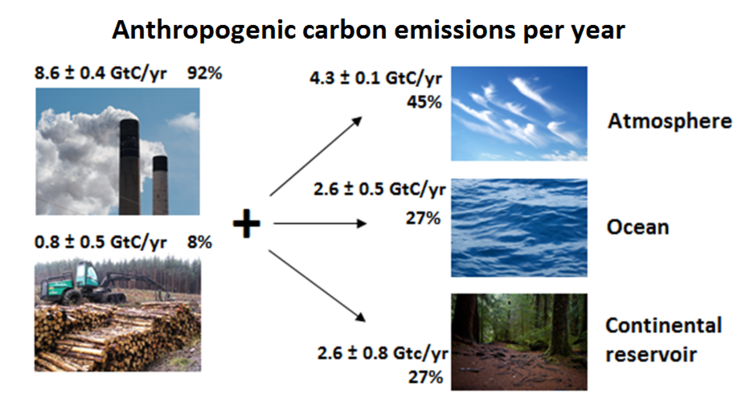
Where do these Emissions Go?
The net destination of emissions from human activities is seen in Figure 2 below. This figure shows the flow of anthropogenic carbon from its emission (dominantly from fossil fuel combustion), to where it ends up, in Earth's major carbon pools. The units are in gigatonnes of carbon per year, portraying the rate of carbon transfer. Remember, a gigatonne is an immense amount of material - equivalent to the mass of 200 million elephants!
References
- ↑ Created internally by a member of the Energy Education team.
- ↑ IPCC, 2012: Glossary of terms. In: Managing the Risks of Extreme Events and Disasters to Advance Climate Change Adaptation [Field, C.B., V. Barros, T.F. Stocker, D. Qin, D.J. Dokken, K.L. Ebi, M.D. Mastrandrea, K.J. Mach, G.-K. Plattner, S.K. Allen, M. Tignor, and P.M. Midgley (eds.)]. A Special Report of Working Groups I and II of the Intergovernmental Panel on Climate Change (IPCC). Cambridge University Press, Cambridge, UK, and New York, NY, USA, pp. 555-564.
- ↑ 3.0 3.1 3.2 3.3 3.4 M. Melieres and C. Marechal, "Warming in the 20th century," in Climate Change: Past, Present and Future 1st ed., U.K.: Wiley, 2015, ch.29, sec.3, pp. 310-312
- ↑ IPCC, The Natural Carbon Cycle [Online], Available: https://www.ipcc.ch/publications_and_data/ar4/wg1/en/ch7s7-3.html
- ↑ Information from Ref.3, images from Wikimedia Commons.

