Absolute zero
Temperature is a measure of how much thermal energy a system has. This measurement means that all of the atoms and molecules which are moving around have a certain amount of kinetic energy (and less obviously potential energy). When all of the molecules (or atoms) in a system stop moving completely, that's as cold as they can get. This temperature, where there's no thermal energy at all, is called absolute zero.[2]
Numerically, this is written as 0 K, -273.15°C, or -459.67°F.
The idea of absolute zero plays into understanding how much energy is available from gas molecules in the ideal gas law, since the temperature has to be measured on an absolute scale (like Kelvin), for the ideal gas law to make sense.[2] Additionally, the idea of absolute zero plays into the physics of blackbody radiation (how much energy radiates out from an object at a particular temperature) and the maximum possible efficiency of a heat engine (called the carnot efficiency).
The concept of absolute zero is also part of the physics of climate change. The average temperature of the Earth, which is about 15°C, would be 288 K. If greenhouse gases increase the temperature of the planet by 1% then it wouldn't go up 0.15 degrees, it would go up 2.88 degrees. Kelvin and Celsius both have the same degree increment but Kelvin is an absolute scale (meaning that it's zero point really is zero) and Celsius is a relative scale (it's zero point is arbitrary - it was chosen by a scientist). This is why the temperature would increase by 2.88 degrees instead of 0.15 degrees. Understanding how these small percent changes in the Earth's temperature can lead to drastic consequences for the planet is an important part of climate science.[3]
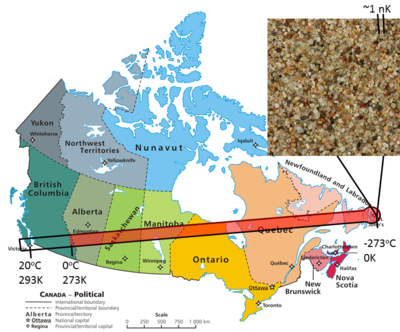
Thermodynamics has shown that it is impossible to get to absolute zero, but physicists have gotten very close.[2] Using laser cooling and magnetic trapping, experiments have been able to get atoms down to temperatures of a few nK (10-9 K) to form Bose Einstein Condensates. To give an idea of how cold this is, imagine a thermometer stretched from Victoria, BC to St. Johns, Newfoundland (7500 km) (see Figure 1).
- 293 K (20°C) Room temperature - downtown Victoria
- 273 K (0°C) Water freezes - the BC-Alberta border
- 0 K - the harbour in downtown St. Johns Newfoundland
- 1 nK - 0.026 mm from the harbour, less than a grain of sand away from the end of the thermometer!
For Further Reading
- Temperature
- Celsius
- Fahrenheit
- Climate
- Thermal energy
- Or explore a random page
References
- ↑ Modified from: By Siim Sepp (Own work) [CC BY-SA 3.0 (http://creativecommons.org/licenses/by-sa/3.0)], via Wikimedia Commons and By E Pluribus Anthony, transferred to Wikimedia Commons by Kaveh (log), optimized by Andrew pmk. (Own work) [Public domain], via Wikimedia Commons by Jason Donev January 12th, 2015.
- ↑ 2.0 2.1 2.2 Winston Smith. "The Importance of Absolute Zero In Science: An Introduction" Accessed Dec.10, 2018. [Online] Available from: https://www.brighthubeducation.com/science-homework-help/111787-importance-of-absolute-zero/
- ↑ Janet Larsen. "Global Temperature" Accessed Dec.10, 2018. [Online] Available from: http://www.earth-policy.org/indicators/C51

