Natural gas formation
Natural gas is a fossil fuel that is composed mainly of methane - composed itself of carbon and hydrogen - and is thus known as a hydrocarbon.[1] Natural gas formation (which is a gas) is essentially the same as the formation of oil (a liquid) and thus takes a significant amount of time with natural gas beginning to form millions of years ago.
The formation of natural gas begins in warm, shallow oceans that were present on the Earth millions of years ago. In these oceans, extremely small dead organic matter - classified as plankton - falls to the floor of the ocean. This plankton consists of animals, called zooplankton, or plants, called phytoplankton. This material then lands on the ocean floor and mixes with inorganic material that enters the ocean by rivers. It is this sediment on the ocean floor that then forms oil and natural gas over many years. The energy in natural gas initially comes from the Sun, and is energy from sunlight trapped by dead plankton.[2]
Formation Process
The process that creates natural gas is the same as the process that creates oil, and is generally the same in most areas. However, there may be different types of plant and animal debris that falls to the ocean floor and slightly different conditions. To form natural gas, the following steps are followed:[2][3]
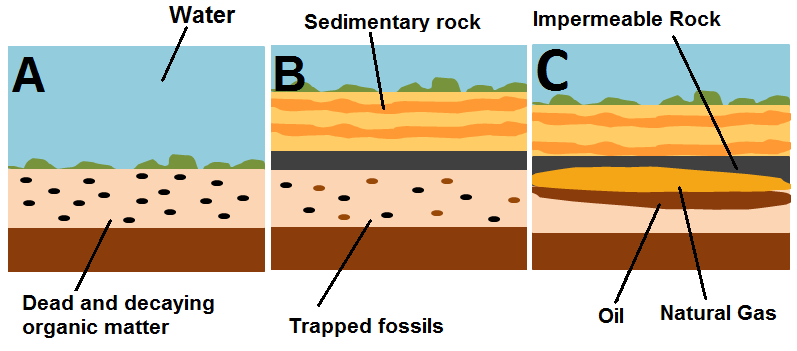
1. Dead plankton - both phytoplankton (including algae) and zooplankton - as well as other soft and hard organic matter tissues (including microbes), sink to the bottom of an ancient ocean and mix with inorganic, clay-like materials that enter these oceans from streams and rivers. This creates an organic-rich mud. This mud can only form in still water environments. This step is shown in Figure 1, panel A.
2. This mud cannot be exposed to too much oxygen, or else the organic matter in the mud would be decomposed by bacteria and disappear quickly. Therefore environments where natural gas can form are known as anoxic environments. Before this organic matter is destroyed, it is buried by more sediment and lithifies (becomes sedimentary rock), creating organic shale. This step is shown in Figure 1, panel B.
3. If this shale is buried between 2 and 4 kilometers, its temperature increases due to its location in the Earths interior. This increasing pressure and temperature of the shale transforms it into a waxy material known as kerogen. Shale that contains this material is known as oil shale.
4. If temperatures of the kerogen are greater than 90°C but lower than 160°C, the kerogen is transformed into oil and natural gas. At temperatures greater than 90°C, the only product is natural gas or graphite.
5. Natural gas and oil are both lighter than water, so as they escape from the source oil shale the products rise through pores in rocks, displacing water. Rock bodies that contain significant amounts of oil or natural gas are known as reservoir rocks. For the gas to remain trapped in the reservoir, there must be some sort of thick, impermeable layer of rock to seal the reservoir. If this seal exists, then oil, gas, and water are trapped beneath and can be drilled into to obtain the oil.[4]
6. Geological changes in the Earth's crust bring these deposits up closer to the surface, making them somewhat easier to access.[5] This step is shown in Figure 1, panel C.
Unconventional Sources
The process above describes the formation of natural gas in a traditional, conventional deposit. However, unconventional deposits such as tight gas, shale gas and coal bed methane are also sources of natural gas. In these deposits, natural gas is stored differently and is also more difficult to access than with traditional deposits.
For further reading
- Tight gas
- Shale gas
- Coal formation
- Hydraulic fracturing
- Or explore a random page
References
- ↑ Natural Resources Canada. (May 28, 2015). Natural Gas: A Primer [Online]. Available: https://www.nrcan.gc.ca/energy/natural-gas/5641
- ↑ 2.0 2.1 R. Wolfson. Energy, Environment and Climate, 2nd ed. New York, U.S.A.: Norton, 2012, pp. 96-97
- ↑ Stephen Marshak. (May 11, 2015). Earth: Portrait of a Planet, 3rd ed. New York, NY, U.S.A:W.W. Norton & Company, 2008
- ↑ Canadian Federation of Earth Sciences. (May 11, 2015). Four Billion Years and Counting: Canada's Geological Heritage, 1st ed. Toronto, ON, Canada.: Nimbus Publishing, 2014
- ↑ J. Kraushaar, R. Ristinen. (May 11, 2015).Energy and the Environment, 2nd ed. Hoboken, NJ, U.S.A.: John Wiley & Sons, 2006, pp. 54